Cell Culture with the CO2 nanomembrane Devices
Cell Culture on the CO2 exchange Devices
The system becomes more complicated with the addition of cells.
Tejas Khire cultured HUVECS in a device that had an inverted design to the one pictured above. (The cells were plated on the top portion (well side) of the nanomembrane with media and the bottom (flat side)of the membrane was exposed to the incubator atmosphere.
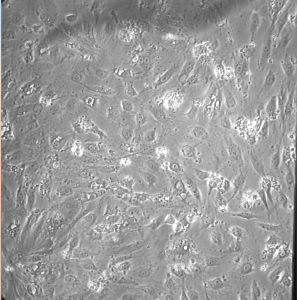
The cells were able to grow for 2 days without media change. One of the things to note is that the cells were able to grow but large vacuoles within some of the cells.
The next thing to try was to grow cells that would be plated onto the glass. The first round of experiments involved the same chamber design given above but with the inner ports directly exposed to atmosphere and the outports will be used to provide media exchange. The setup could be used to provide fluid flow to the system, but for the initial tests the system was kept under static conditions. A representative image of the system is shown below.
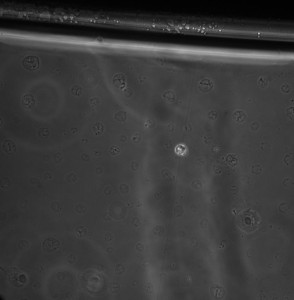
For the first round of experiments. Cells were seeded into the nanomembrane at a low density and allowed to grow with the membrane, providing the only interface for the cells to the atmosphere. The media was changed every 2 days for the first round of experiments. However, the cells were all dead on the fourth day. Image of the cells is given below (day 4).
The media was changed every two days because if the media is changed the following day the cells are promptly washed away. This is likely due to inadequate matrix formation on the coverslip. Pretreating the surface with a substrate/chemical (fibronectin, aptease(sp?), serum) will most likely improve adhesion onto the glass.
Other things to note is that the media “appears” white while in the chamber.
But upon replacement of the media, the old media at the outlet is actually a slightly more yellow color than the replacement media (inlet)
It seems that the media is properly buffered, but that there is a depletion of nutrients or build up of waste in the system. Both of these limit cell growth. However, it is difficult to say exactly.
Cell Culture on the CO2 exchange Devices Continued
Following these experiments cells were seeded in similar numbers to the first experiments. However, the difference is that the bottom chamber where the cells were seeded was increased. This allows for more media in the system, but at the same time the amount of distance the CO2 has to travel to buffer the system is increased.
For reference the initial volume of the first chamber was 1.4mm^3. (1.4microliters)
The new chamber designs were 12.6mm^3 (12.6microliters) and 8.4mm^3 (8.4microliters). For reference the membrane surface area is about 1.4mm^2, surface area of a pinhead is about 2mm^2.
Following the initial setup the media was changed on the second day and then each day subsequently at the same time. However on the fourth day the cells were not viable once again. The picture below was taken on the fourth day.
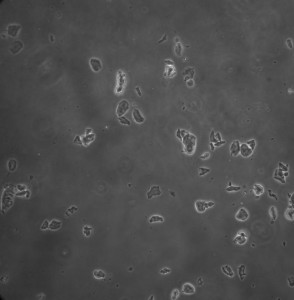
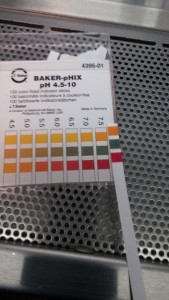
This time around the pH was taken on the third day, following a media change. The pH was measured using pH strips (far right on picture below).
The cells seem to be only slightly basic. Cell death is again attributed to depletion of nutrients or build up of wastes. Only the larger chamber design was tested due to failure of the other designs(membrane breaking).
If media is just added to the device with dead cells, the media remains pink and the media in the tubes is white. This is clearly seen on the larger chip designs.
Conclusion
The nanomembranes are able to provide adequate CO2 exchange to the system as evidenced by the first sets of experiments. The media does not leak onto the top of the membrane and CO2 enters the media in a predictable manner.
When cells are added the metric of success is a little harder to define. The cells are able to grow but may require more media changes per day as the number of cells increase. The volumes are so small that nutrients like glucose are rapidly depleted and it is possible that waste also builds up to toxic levels. However, the “lab-on-a-chip-on-air” and the original design with cell provide adequate CO2 buffering, temperature and sterile environment for cell culture.
The question of minimum amount of media required for cell culture remains unknown at this time.
Future Work
All the cell culture experiments should be repeated for accuracy.
Cell culture with addition of a substrate.
Accurate modeling of CO2 exchange in a system.
Fluid flow on the cells for aligned monolayers
Studies on when media should be changed.
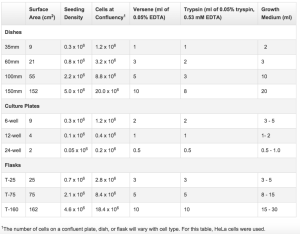
It is unlikely that results from “large” cell culture could be scaled down but life technologies provides some numbers.
http://www.lifetechnologies.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/cell-culture-useful-numbers.html
But assuming linear trends the media required for the surface area is 20 microliters. So we might be within a reasonable range?
Repeat experiments from lab-on-a-chip-on-air