bEnd3 culture under flow for BBB mimetic (sort of)
Hi all,
For the past one month I have been working on a height-based device for culturing bEnd3 (mouse brain endothelial cells) under flow. Ideally, this device is to constitute a blood brain barrier (BBB) mimetic.
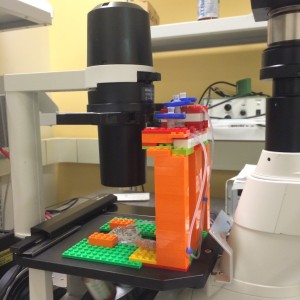
Device Full View (Front):

The fluid flow is driven by hydrostatic pressure, created through the differences in the heights of the fluid columns. The height differential between the left and the right syringe drives the flow on the top side of the membrane where the bEnd3 were seeded. The middle syringe is connected to the bottom side of the membrane. The height of the fluid column in the middle syringe is an average of those in the left and the right syringe. In theory, such a fluid column height would create a back pressure that evens the total transmembrane pressure to zero.
Device Full View (Back):

Note that eventually the heights of the fluid columns would all be equal over time. To prevent this from happening, a peristaltic pump is used to dump the fluid built-up in the right syringe back to the left syringe. The long silicone tubing shown is used for the peristaltic pumping. Note:If the height differential creates a flow much faster than that enabled by the peristaltic pumping, the height differential would eventually reduces to match the peristalsis.
Assembled flow cell containing the SiN membrane chip:
The cells were first grown under an open, static condition (for details refer to this post). At near confluence, a lid (with 4 ports) was place on top to convert the open slit on top of the membrane to a closed channel. The whole assembly is held together through clamping (via screws).
Below are additional views of the device:
In the incubator:

On the microscope:
bEnd3 Culture:
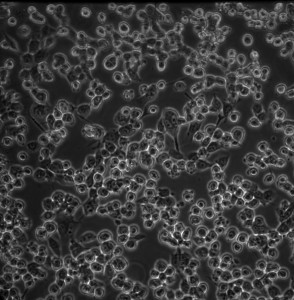
Day 0 (closed system assembled): After assembling the system, I set the flow to ~ 400 uL/min (shear stress ~4.44 dyn/cm^2). Initially though the shear stress is much higher, because I am using the fluid volume of the left syringe to fill the right syringe. Some cells may have sheared off because of this, but an appreciable number remain attached. I notice that the cells were also rounding up a bit, possibly because the system is at room temp for too long. I notice that this tends to happen for bEnd3, especially when the they are not at confluency.
Day 1 :
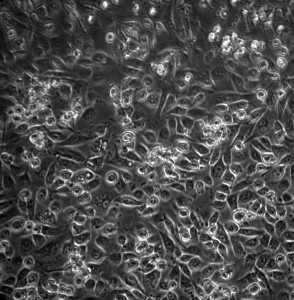
I always worried that a height-bases system is more susceptible to flow cloggage, If there is a bubble the surface tension may be high enough to stall the flow (since the pressure created through the height difference is actually pretty low). This fear did manifest. There was a small bubble and the flow did stall. So no cell alignment observed, but the cells were able to proliferate more to achieve near-confluency. I debubbled the system and resumed the flow, this at higher flow of 800 uL/min (shear stress ~8.88 dyn/cm^2). With the higher flow rate there should be less chance for flow stalling.
Day 2:
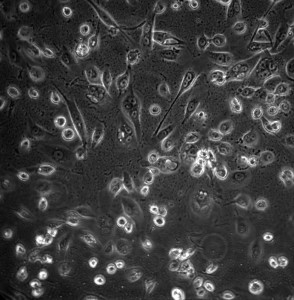
No flow stalling, but an appreciable number of cells were sheared off. Some of the adherent cell exhibited a more spread and elongated morphology. Unfortunately, they seem to elongate perpendicular to the flow (which is from right to left, not up and down). I decided to renew media since it’s been 2 days.
After renewal i may have lost more cells:

Many also rounded up. In the future I will manually set the fluid height differential rather than letting the system do it itself (since that will create higher shear stress). Since i did not observe cell alignment, I upped the flow rate to 1.2 mL/min (13.33 dyn/cm^2) and decided to leave the system alone for 3 days (so the cells don’t subject to temperature shift too often, plus I have new media anyway so it should be good for some time).
Day 5:
Bummer… no idea what happened exactly. The cells were super spread. Also not sure if they are alive. There are also a lot of streaks. These streaks may either be debris left behind as the cells sheared off or contaminants (I did renew media 3 days ago). If they are contaminants though, they are probably something other than bacteria, since they are more than cell-sized (these spread cells are much larger than the normal endothelial cells).
COMMENT: In retrospect, I should have kept the flow rate low first to make sure the cells remain adherent. After all, viability should be the top priority. For the next iteration I will let the bEnd3 be at confluency for at least 2 days before starting the flow. It is unclear what happened between day 3 and day 5, but hopefully this would be avoided on the revisit.
I also found recent publications (2014 and 2015, from the same group) that the brain endothelial cells (human brain microvascular endothelial cells/HBMECs) actually resist alignment in the direction of flow (and prefer to stay perpendicular to the flow). So perhaps I should not expect flow alignment in the first place.


That last image does look like contamination. There seems to be very few cells and a film between them. Also some really dead looking cells with blebs.
I see. Will try to keep it koshered next time.