Discoloration – oven vs. incubator
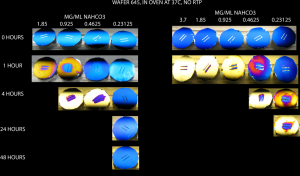
A couple weeks ago, I posted some pictures that showed a non-linear “dose-response” curve for NaHCO3 and discoloration in the incubator (post). I repeated this experiment at 37C in the benchtop oven.
These pictures show that there was a NaHCO3 concentration dependence on the discoloration rate in the oven. In the 2nd set of experiments (the right half of the image), discoloration was faster because I messed up when making the NaHCO3 stock solution. So in the oven, discoloration from NaHCO3 was faster and it followed a concentration dependence.
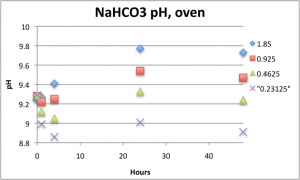
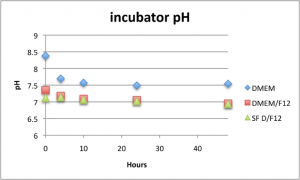
Here is how the pH changed with time in the oven (top) and incubator (bottom).
In the oven, the pH of all solutions either didn’t change or increased over 48 hours. This is in stark contrast to the pH in the incubator, which decreased to physiological range within 3 hours. Higher NaHCO3 concentrations yielded higher pH’s for each condition.
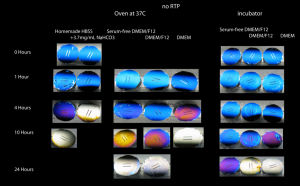
This result motivated a study in which I looked at discoloration in the oven at 37C and in the incubator with DMEM and DMEM/F12 cell media.
After 10 hours at 37C in the oven, chips in DMEM, DMEM/F12 and serum-free DMEM/F12 were discolored, or at least well on their way to complete discoloration. After 10 hours in the incubator, however, only the chip in DMEM showed discoloration. In the oven, discoloration with serum-free DMEM/F12 was complete at 24 hours but had only just started in the incubator.
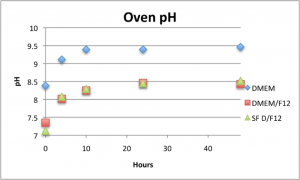
Like the bicarbonate, cell media pH increased slightly in the oven, but pH slightly decreased in the incubator.
Although it isn’t a huge effect, something about the incubator helps to slow down discoloration rates in bicarbonate solutions and cell media. Since the temperatures are the same in these experiments, it must be the pCO2 (~5% in the incubator vs. <0.1% in air).


Yes. It certainly seems that outside the incubator we can think of the effect as merely pH driven, but that something more complex is going on in the incubator that makes bicarbonate-based media more aggressive even at the same pH.