Gold separations with carbonized membranes
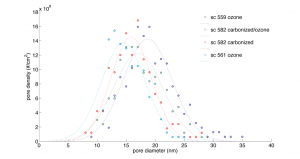
Recently, I have been trying to pass 5 nm, 10 nm, and 15 nm gold through untreated, carbonized, and ozone/carbonized membranes. These experiments were done in the pressure cell with fresh samples. I used a 1:1 dilution of Au stock and DI water. To wet the membrane I placed a 40 uL drop of water on the front-side. I have accounted for this dilution in my absorbance calculations shown below. I let each experiment run until at least 100 uL had passed. Here is a comparison of the three membrane distributions I looked at:
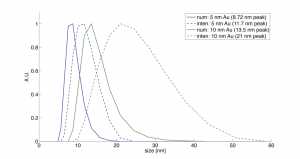
I wanted to check the size distribution of the gold so I used the Malvern to measure a 5 nm and 10 nm solution.
It would appear that both the 5 nm and 10 nm gold are actually 1.5x larger than specified according to DLS. The full width half max (FWHM) of the 5 nm and 10 nm solutions are 6 nm and 8 nm, respectively.
ozoned sc 559
The top plot shows the absorbance data (adjusted for water absorption and dilution). The bottom plot shows the pore distribution (blue) and % concentration passed (I took the ratio of the 1-1 Au peak and filtrate peak at 525 nm). The horizontal error bars reflect the FWHM of the gold distribution based on the Malvern measurements. I estimated the 15 nm Au distribution to be the same as the 10 nm.
carbonized/ozoned sc582
carbonized sc582
ozoned sc 561
These results indicate that we can achieve a sharp separation of gold using membranes that are carbonized and carbonized/ozone treated. The next step is to understand why our effective cut-offs are so much lower than the apparent cut-off. Carbonization is assumed to neutralize the negative charge on the pore walls, so why does 5 nm not pass through? How does the combination of ozone and carbonization affect Au transport?