NSF proposal on Catalytic Membrane Reactors
I submitted a proposal last week with Tom & I as co-PI’s to NSF’s Process Reaction Engineering division in CBET.
Novel Catalytic Membrane Reactor Based on Ultra-thin Palladium Film Supported on Nano-structured Inorganic Membrane
I’m attaching the intro. paragraph and a couple of the figures to give a general idea of what the proposal is about.
We propose developing a new class of hydrogen perm-selective composite membranes by using our novel nano-porous membrane material as a substrate to support an ultra-thin (<100 nm) palladium (Pd) film and applying this composite membrane in the direct hydroxylation of benzene to form phenol. The nano- membrane material, termed porous nanocrystalline silicon (pnc-Si), was developed previously in our lab and has nominal thickness of less than 50 nm and tunable pores in diameters ranging from 10 to 40 nm. It has been applied successfully in a range of high-resolution molecular filtration demonstrations where it shows record level fluid throughput and high sieving precision. In contrast to other nano-porous membrane materials that require prohibitively expensive fabrication techniques such as neutron bombardment or focused ion beam milling, the pnc-Si material is created with standard semiconductor fabrication processes that are scalable. Here, we want to apply the pnc-Si membrane to catalytic reactors. The pnc-Si membrane possesses several unique characteristics that make its application in catalytic membrane reactors highly compelling including: nm-scale thickness and porosity, tunable pore size, smooth surface morphology, and the ability to coat or functionalize surface to stabilize a thin palladium film. We believe it will be possible to
achieve stable Pd (or Pd-alloy) films that
are at least ten-fold thinner than what can
typically be obtained with state-of-the-art
Pd membranes today that are supported on
porous ceramic frit materials, which have
surface morphology features on a scale of
several 100s of nm to microns. The
morphology of pnc-Si makes much
thinner, defect-free films possible, and the
nm-scale thinness ensure high gas
diffusion rates (Figure 1). Reduction of
Pd film thickness is critical both from the
standpoint of hydrogen permeation and
usage of the precious metal. We envisage
a range of important application areas for these ultrathin membrane reactors related to the perm-selectivity property of Pd towards hydrogen including hydrogen production, purification, hydrogenation, and hydroxylation. Specifically, in this proposed effort we aim to develop and optimize fabrication techniques for creating the ultrathin Pd membrane, measure its perm-selectivity towards hydrogen, and optimize its use in an industrially important reaction (direct hydroxylation of benzene to phenol). Preliminary work in our lab exploring deposition of a palladium-silver alloy through electron-beam evaporation has shown promising results for obtaining good wetting of the pnc-Si surface. If successful, this research could transform the field of palladium-based catalytic reactors for a host of industrially important applications.
Figure 1: Scanning electron micrographs that illustrate the contrast in substrate morphology and resulting Pd film thickness. (A) Cross-section near outer surface of typical porous ceramic tube with a thin (~4 μm) Pd film coating (from [17]). (B) Edge of 15 nm thick pnc-Si membrane with a 100 nm Pd film on its bottom surface. (Many tanks to Josh W. for the help with this!)
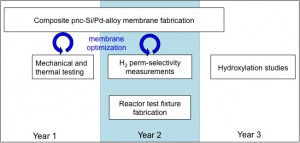
Figure 2: Project timeline.The motivation for this is to develop a direct hydroxylation route for making phenol from benzene by reacting a mixture of air and benzene on one side of the membrane with hydrogen that permeates through. It is dangerous to premix hydrogen w/ air and then pass it over a catalyst, plus it’s generally difficult to control the reaction selectivity. By introducing the hydrogen via the perm-selective membrane, one has better control over the reaction. The present method for phenol manufacture involves cumene and cumene hydroxide intermediates. The process is energy intensive, has a low per-pass yield, a dangerous intermediate, and creates an excess of acetone by-product. Some folks have shown promising results using a “conventional” palladium membrane that is a couple microns thick supported on a porous ceramic tube (see figure below). The argument I make is that by using pnc-Si as the Pd film substrate, one can obtain mush thinner Pd films. This saves on the expense of Pd, and enhances the mass transport of hydrogen to the reaction.
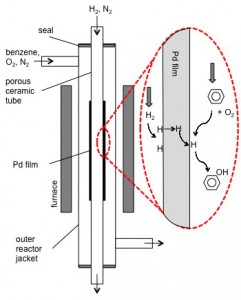
Figure 3: Schematic of test set up and reaction scheme for direct hydroxylation of benzene to phenol using a hydrogen perm-selective Pd membrane reactor. Reaction conditions are typically ambient pressure, 150-200 ̊C, and molar flow ratio of H2, O2, and benzene of 5:1:0.026.