Sphingosine-1-phosphate: Quick Introduction and TEER Results
Introduction
Sphingosine-1-phosphate: production and endothelial cell signaling
Sphingosine-1-phosphate (S1P) is a bioactive sphingolipid produced through the phosphorylation of sphingosine, a component of the plasma membrane, by SphK1/2. Many cells produce and store S1P, the largest source of which is platelets, due to their high native SphK activity. Erythrocytes and leukocytes are also sources of S1P in the blood, but to a significantly less degree then platelets. Non-blood cells, including vascular endothelial cells and hepatocytes can also produce and release S1P. In circulation, S1P is primarily bound to HDLs or albumins, allowing for increase half-life. While S1P is well known for its role in lymphocyte trafficking, we are particularly interested in its ability to maintain or regulate endothelial barrier properties. Extracellular S1P can bind endothelial cell surface expressed g-protein coupled receptors (S1P1/3) to enhance or decrease barrier properties. This is mainly done through the regulation of TJs and adherens junctions, like VE-cadherin. S1P signalling through it’s third receptor was also found to mobilize p-selectin on the endothelial cell surface, enhancing leukocyte rolling (Nussbaum et al., Nat Commun 2015).
Apicobasal polarity, S1P receptor expression, and immunity
In the theme of endothelial cell apicobasal polarity, one important aspect of S1P receptor expression is it’s ability to be differentially expressed on apical versus basal sides of vascular endothelial cells (Cannon et al., PNAS 2012). This leads to the hypothesis that apical or basal delivery of S1P may differentially regulate endothelial cell barrier properties. The conclusions of this hypothesis may open the door to polarity specific drug development.
Methods
Human umbilical vein and human pulmonary microvascular endothelial cells were cultured on PC transwell membrane inserts with 1 um pores. TEER was monitored until peak resistance was observed (Figure 1). At peak resistance, media supplemented with 1 uM BSA-conjugated S1P was added to the apical or basal chamber, and TEER was continuously monitored over 30 mins. The setup for continuous TEER monitoring can be seen in Figure 2.
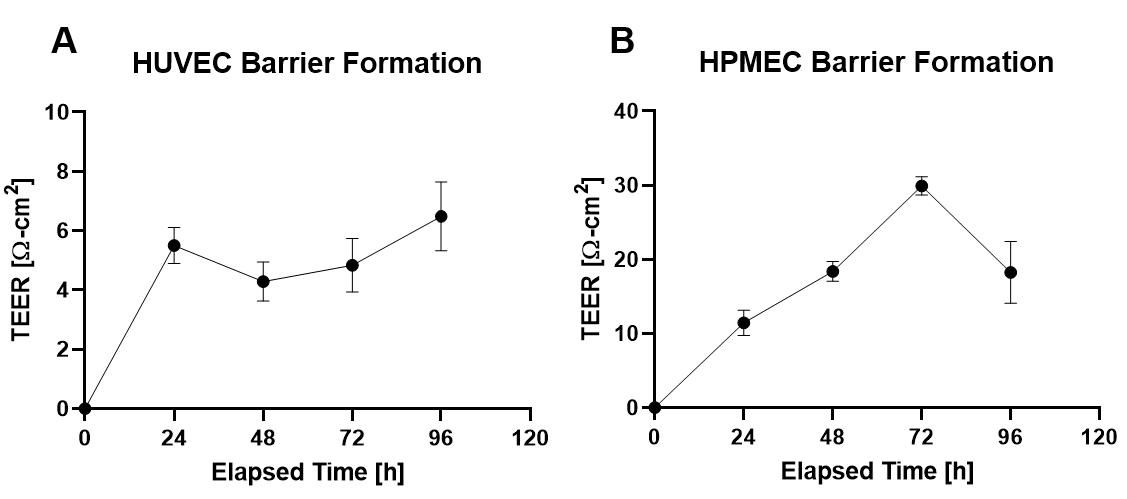

Results
Luminal and abluminal delivery of S1P seem to have similar effects of endothelial cell barrier properties. Interestingly, these effects appeared barrier protective in HUVECs and barrier disruptive in HPMECs (Figures 3&4).
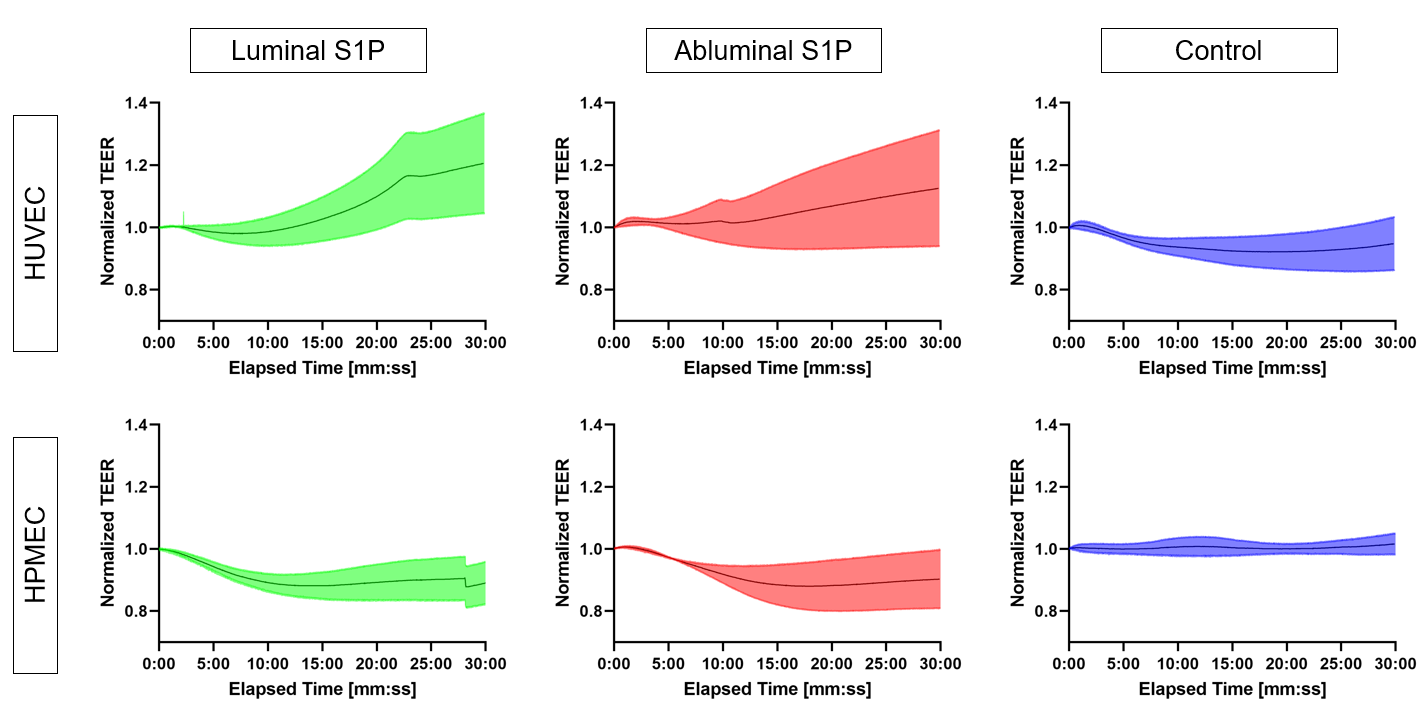
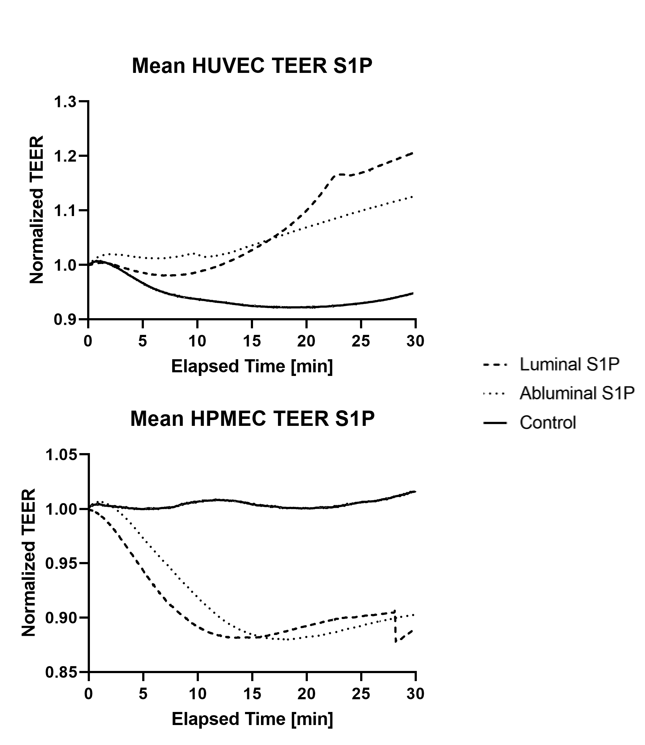
Conclusions
S1P appears to have variable effects of EC barrier properties. Given the literature that establishes this paradox suggests a role of different receptors, our next goal is to characterize receptor expression on these two cell types and compare. We would also like to better understand how these changes in barrier properties may regulate neutrophil trafficking.