Discoloration – Buffers and pH
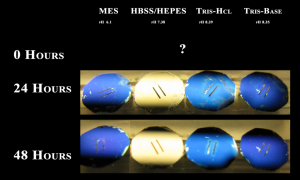
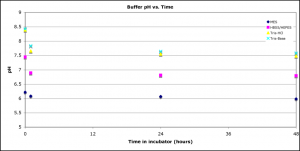
To further investigate the mechanism of discoloration, I prepared buffers at different pH’s and measured discoloration and pH over time. Buffers were: 0.1M MES, pH=pKa=6.1; 0.1M Tris-Base, pH=pKa=8.39; 0.1M Tris-HCl, pH=pKa=8.35; 10mM HEPES in 1X HBSS, pH 7.38, where HEPES pKa = 7.31. This was done in the incubator.
Experiment 1:
Experiment 2:
pH vs. Time:
In both experiments, only HBSS with10mM HEPES caused discoloration, even after 4-5 days in the incubator. Sorry – I lost the day 0 pictures for Experiment 2. Even though the Tris buffers were relatively basic at each time point, these chips did not discolor, which suggests that pH is not the determining factor for discoloration.
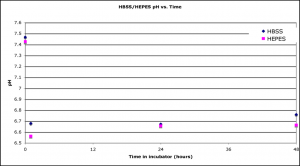
The above two experiments showed that HBSS with 10mM HEPES caused discoloration. What was different about this solution? HBSS with 10mM HEPES is the only solution that contained non-buffer salts (KCl, NaCl, KH2PO4, Na2HPO4, Dextrose and our old friend NaHCO3). I separated this solution into 10mM HEPES, pH 7.47 and HBSS, pH 7.35 and followed discoloration and pH over time.
Here, there was a rapid decrease in pH after adding the solutions to the incubator, but then pH stabilized. The HBSS solution rapidly discolored, but 10mM HEPES did not discolor, even out to day 4. Again, since the pH of these 2 solutions were essentially the same throughout the experiment, this result suggests that pH is not the sole mechanism for discoloration.


RTP treatment for these samples? Which wafer?
I guess I am convinced that it is not only or necessarily pH.
Excellent work.
What is next? Can you check out the effects of HBSS components or are there too many? If its bicarbonate, what is so special about this molecule? Are there other similar molecules that we can test?
Tom – no RTP; w638 for top figure, w606 for bottom figure
Jim – Thanks, I’m on it. Should be posted soon.