Adsorption Negative Assay
If protein is adsorbing to a surface, it’s leaving the solution. We can indirectly measure how much protein is leaving the solution, and that may tell us something about how much these surfaces are adsorobing.
For my first attempt, I used the Bradford Reagent to measure the concentration of the solution after protein has remained on the surface for 30min.
Here’s how I set up this assay:
- Apply 30uL of .5mg/mL BSA to sample (membrane chip, pes, or cellulose).
- Remove 20uL of solution after 20min.
- Dilute in 1000uL of Bradford Reagent (1:5 from stock).
- Measure absorbance on spec after 10min.
Here’s the results:
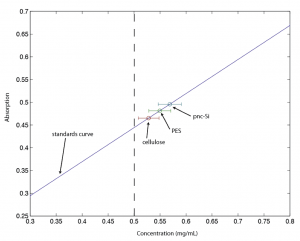
You can see that the calculated solution concentrations are actually higher than the starting concentration (dotted line). This is either due to handling error or inherent error in this setup. The standard curve was set up in the same manner as the samples.
Even if I can make the points appear in a reasonable place on the curve, there’s a good deal of overlapping error and little difference between pes, cell, and pnc-Si. I’ll continue to mess with this a little more, first by trying different concentrations of starting sample (which may allow for more observable differences).