Discoloration – HBSS components
In my last discoloration experiment, I tested buffers with different pKa’s (MES, HEPES, and Tris). I found that only 10 mM HEPES in HBSS caused discoloration, and furthermore, that the HBSS component of this solution was the culprit (post). Fortunately, HBSS is a rather simple solution; it only consists of KCl, NaCl, Glucose, KH2PO4, Na2HPO4 and NaHCO3 (formulation sheet). This simplicity allowed me to separate HBSS into its components and test for discoloration. I had a hunch that NaCl, KCl and glucose at these concentrations wouldn’t cause discoloration. So, I combined NaCl and KCl into one solution and added NaH2PO4. I also added Na2HPO4 to glucose. I tested KH2PO4 and NaHCO3 alone. Of course, all of the concentrations that I tested matched those in HBSS. DMEM/F12 was used as a negative control. This was done on wafer 625 without RTP.

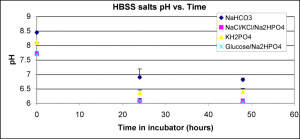
I also tracked the pH of these solutions over time.
Even though NaHCO3 pH was higher than the other solutions over time, there was no discoloration. In fact, none of these solutions caused discoloration, even though combined as HBSS they did cause discoloration. This is the first time (I think) that NaHCO3 didn’t cause discoloration, although it’s about 10x less concentrated than NaHCO3 in DMEM.
Do you buy HBSS or mix it yourself? Did you also run HBSS as a control simultaneously with these solutions?
If you buy HBSS, I would try making it yourself to see if you can reproduce the effect. Purchased solutions are always a little different, in my experience. It seems odd that mixing these solutions together kills the membranes while subsets of them do not. Perhaps you could try removing one at a time to see if you can gather more details?
I really find it hard to believe that the explanation is as complex as it appears to be from the experiments completed thus far. The root cause is a highly electronegative ion interacting in some way with the Si surface – period. Typically, this is OH, but fluorine, and any ion containing oxygen could also be involved. It just has to create a transient bond with Si, pull electrons away from the Si, and weaken the other bonds for chemical attack. Obviously, whatever carries away the Si must also be soluable, or solulizable by a secondary reaction….
When you try to reconstitute the bad mix in pieces I would suggest adding the salts to bicarbonate as one test. We’ve seen these components discolor membranes separately at higher concentrations (as I recall) so I’m curious if their effects are additive or worse.
Thanks for the comments. This HBSS was bought from the Bio Supply Center. Since it’s a simple solution of salts, I made up the components and then made a batch of “homemade” HBSS. The experiment with the homemade HBSS and different combinations of the components was started on Friday.