Development of Arising Retinal Pigment Epithelium-19 (ARPE-19) Barrier Tissues on NPN
Author: Kevin Ling
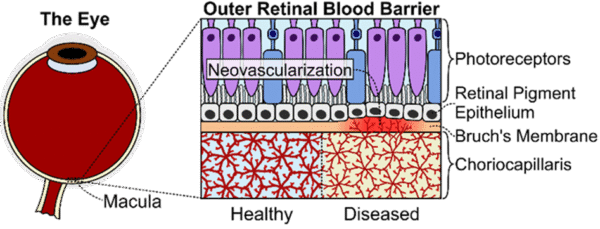
Dysfunction of the outer retinal blood barrier (oRBB), which is composed of the retinal pigment epithelium (RPE), Bruch’s membrane, and choriocapillaris (CC), is implicated in age-related macular degeneration (AMD), which causes irreversible central vision loss [1-4]. AMD is the leading cause of blindness in industrialized countries; it is estimated that by 2040, about 288 million will be affected by AMD globally [5-7]. AMD is classified as “wet” or “dry” based on the presence (wet) or lack of (dry) blood vessel infiltration through the Bruch’s membrane from the choroid [1, 2, 4, 8-10]. While some effective treatments for wet AMD exist, there are no clinically proven treatments for dry AMD [8, 10, 11].
Animal models for the oRBB and AMD are severely limited. Mouse retinae have numerous structural differences compared to human retinae: they lack maculae, have different photoreceptor distributions, have thinner Bruch’s membrane, and their RPE has a higher relative phagocytic load [12]. The retinae of non-human primates, while physiologically similar to those of humans, are impractical for mechanistic or therapeutic studies due to genetic inflexibility, slow disease progression, and high cost. Moreover, primate AMD models still do not recapitulate all features of human AMD [13-15]. Hence, there is a critical need for improved models of the oRBB for pre-clinical ocular disease investigations and pharmacologic development [16].
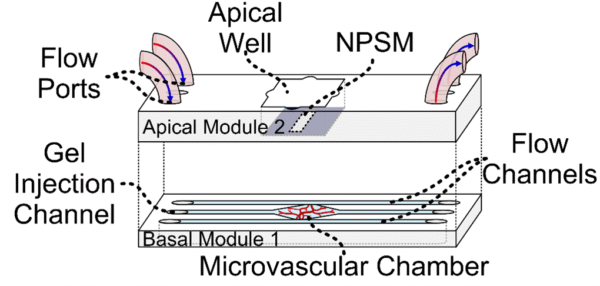
Hence, we sought to utilize the advantages of nanoporous silicon nitride membranes (NPN) and the µSiM device to develop an improved model of ARPE-19 barrier tissues. Specifically, these technologies confer improved imaging capabilities and opportunities to study oRBB tissue development and crosstalk between RPE and the basally situated CC in vitro compared to conventional transwell cultures [17, 18]. To that end, we designed the novel outer retinal blood barrier-on-a-chip (oRBB-C), which serves as a platform to enable the interrogation of the interface between apical situated RPE tissue mimetic and basally situated microvascular networks (Fig. 1).
 |
|
 |
|
 |
 |
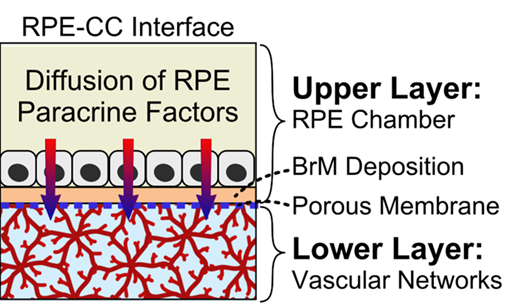
| Fig. 1: A) Organization of the oRBB. Key signs of AMD disease progression include drusen deposition in the Bruch’s membrane, alterations in CC flow patterns, aberrant neovascularization from the choriocapillaris, and RPE/CC/photoreceptor degeneration, but the precise etiology of AMD remains poorly understood [1, 2, 8-10, 19-22] B) outer retinal blood barrier-on-a-chip (oRBB-C) schematic. Endothelial cell derived microvasculature will be developed in Module 1. RPE barrier tissue will be cultured in the apical well of Module 2. Module 1 channels connect to flow/gel injection ports on Module 2. Modules 1 and 2 are separated by NPN. C) RPE-CC interface in oRBB-C. In oRBB-C apical Module 2, RPE cells will be seeded atop an NPSM and juxtaposed to the microvasculature networks in oRBB-C basal Module 1. NPSM will enable RPE to form a robust monolayer with functional barrier properties, while also allowing the paracrine factor exchange between both tissue layers. D) photograph of a fully assembled oRBB-C. | |
In this blog post, we will share updates regarding the development of ARPE-19 barrier tissues in the apical well of oRBB-Chip which recapitulate canonical morphological and functional characteristics of this cell type.
Methods
oRBB-chips with nanoporous silicon nitride membranes were pre-treated with laminin for 24 hours prior to cell seeding. Arising retinal pigment epithelial cells (ARPE-19, ATCC) were seeded in the apical well of oRBB-chips at a density of 50,000 cells/cm2. For the first six days after seeding, a growth media consisting of Dulbecco’s modified eagle medium: nutrient mixture F12 (DMEM/F12) with 10% fetal bovine serum (FBS) and 1% pencillin/streptomycin (PS) was used to replace the growth media of ARPE-19 oRBB-chip cultures every other day. Starting on day seven, growth media was replaced with a modified differentiation media, consisting of DMEM/F12 with 1% FBS and 10 mM Nicotinamide based on a protocol described by Hazim et al. [23]. The µSiM transepithelial electrical resistance (TEER) module developed by Mehran et al. the Rochester Institute of Technology was used to measure TEER immediately before cell seeding to establish a baseline value and on the final day before fixation of the culture [17]. The sampling permeability method, described and developed by McCloskey et al., was using to assess the permeability of ARPE-19 oRBB-chip cultures to fluorescent lucifer yellow [18]. oRBB-chip controls, which only received laminin coatings, were used to establish a baseline for both TEER and permeability measurements. After fixation of oRBB-chip devices with 4% paraformaldehyde, oRBB-chip cultures were stained with primary, secondary, and pre-conjugated antibodies for immunofluorescence confocal microscopy.
Transepithelial Electrical Resistance
Transepithelial electrical resistance (TEER) is a quantitative metric that is used to characterize the combined paracellular and transcellular resistance of epithelial tissue layers to electrical current. This metric can be correlated with the integrity of tight junctions and hence is often used to characterize the barrier function of RPE [17, 24-26]. TEER analysis of ARPE-19 oRBB-chip cultures show that over a period of 28 days, systemic TEER (TEER of the entire device) is increased after 28 days of culture with ARPE-19 cells (Fig. 3A). Our results show that the systemic TEER (TEER measured for the entire oRBB-Chip device) increases over a period of 28 days of culture (Fig. 3). The average cell TEER (device TEER subtracted from systemic TEER) measured for ARPE-19 cultured in oRBB-chip devices was 136.6 ± 20.9 Ωcm2, which is higher than the expected value of 42 ± 7 Ωcm2 based on recent literature [23, 25, 27, 28] (Fig. 3). This may possibly be a result of using NPN rather than traditional microporous membranes found in conventional transwell cultures, in which the literature values were derived. Unexpectedly, control devices which received laminin coatings but did not receive cells and underwent the full 28 day culture period (with all media changes), also had increased TEER values despite lacking any ARPE-19 cells (Fig 3).
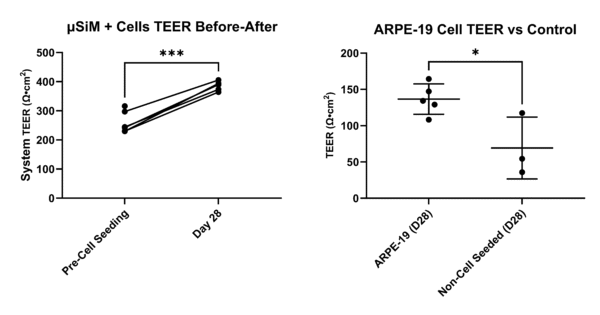 |
| Fig. 3: Left: Systemic TEER over time. Total oRBB-chip TEER increases over a period of 28 days of culture. Right: ARPE-19 cell TEER. Cell TEER is higher compared to laminin treated, non-cell seeded controls. Statistics: Statistics: ANOVA with Tukey’s Multiple Comparison’s test, ****p<0.00005, ***p<0.0005, **p<0.005, *p<0.05. |
Sampling Permeability
Small molecule permeability was also measured to assess the barrier functionality of ARPE-19 tissues cultured in oRBB-chips. While in vivo estimates of permeability of ARPE-19 have been elusive, it is estimated that the permeability of the outer retinal blood barrier is similar to that of the blood brain barrier, around 1.5 x 10-3 cm/min. [18, 29]. Our results show that the systemic permeability (small molecule permeability of the entire oRBB-Chip device, including cells) decreases significantly after 7 days of culture during the proliferation period, and decreases more over the full 28 day period (Fig. 4). 10mM Nicotinamide treatment also caused decreased permeability of ARPE-19 compared to control devices, which received standard ARPE-19 growth media.
 |
| Fig. 4: Left: Systemic permeability. Total permeability of oRBB-Chip ARPE-19 cultures decreases after 7 days, and once again over a period of 28 days of culture. Right: ARPE-19 cell permeability. The sampling permeability of ARPE-19 cells in cultures treated with 10 mM Nicotinamide media supplementation is significantly lower than cultures which received standard ARPE-19 growth media. Statistics: ANOVA with Tukey’s multiple comparisons test, ***: p < 0.0005, ****: p < 0.00005; ANOVA |
Key ARPE-19 Markers are recapitulated in the oRBB-Chip
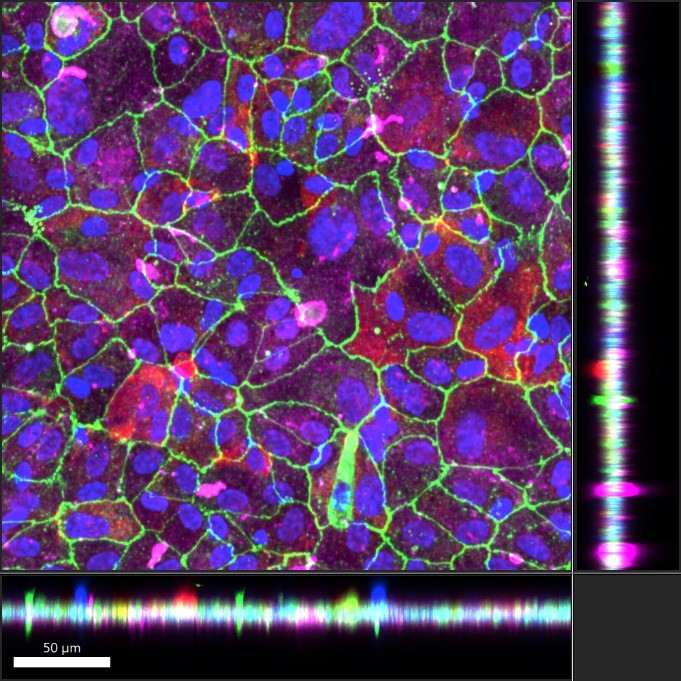
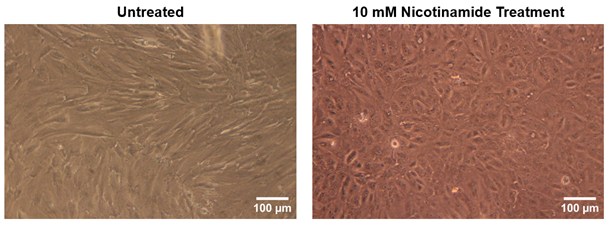
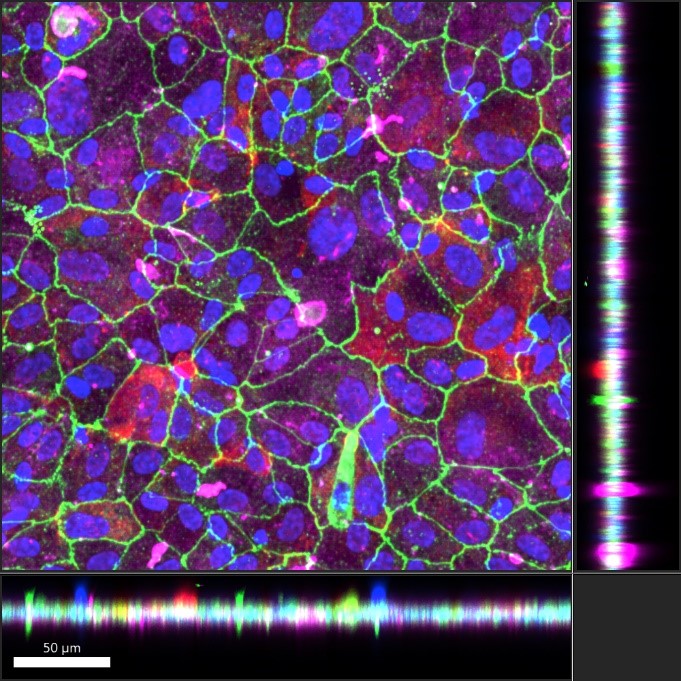
Immunohistochemistry was also utilized to study the morphological development and maturation of ARPE-19 cultures in oRBB-chip devices. Key indicators of ARPE-19 maturation include canonical apical localization of Ezrin, a structural protein and the expression of tight junction protein zonula occludens-1 (ZO-1) [30, 31]. ZO-1 was also used to determine if ARPE-19 cells formed tight junctions and hexagonal cobblestone packing morphology. We found that in ARPE-19 oRBB-chip cultures which did not receive Nicotinamide, ARPE-19 developed fibroblast-like morphology, indicating an undesirable epithelial to mesenchymal transition [23, 32]. ARPE-19 oRBB-chip cultures which received 10 mM Nicotinamide supplemented media developed barrier tissues with higher expression of Ezrin, ZO-1, as well as hexagonal cobblestone packing (Fig. 5). Orthogonal projections of confocal images show that Ezrin is apically localized relative to cell nuclei (Hoechst), ZO-1, and Collagen IV, and that Collagen IV deposition is basal (Fig. 6). This was surprising, given that these cultures were not given specific apical-basal polarization cues (i.e., basal VEGF). It is possible that pre-treatment of the membranes with laminin served as a polarizing cue for ARPE-19 oRBB-chip cultures.
 |
 |
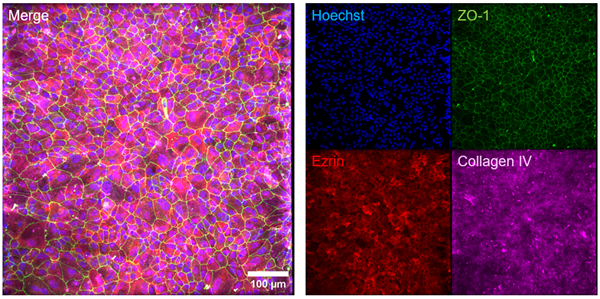 |
| Fig. 5: Top: Nicotinamide rescues ARPE-19 differentiation and maturation. ARPE-19 oRBB-chip cultures which received 10 mM Nicotinamide supplemented media had higher ZO-1 and Ezrin expression, and the characteristics hexagonal packing for ARPE-19 barrier tissues was clearly developed. Bottom: Collagen IV, a protein that is found in native Bruch’s Membrane, was also present in oRBB-Chip ARPE-19 cultures after 28 days of culture. Because Collagen IV was not included in the initial pre-treatment of NPN prior to cell seeding, this is an indication that ARPE-19 barrier tissues are able to generate and deposit basement membrane proteins in the oRBB-chip model. Scale bars: 100 μm. |
 |
 |
 |
|
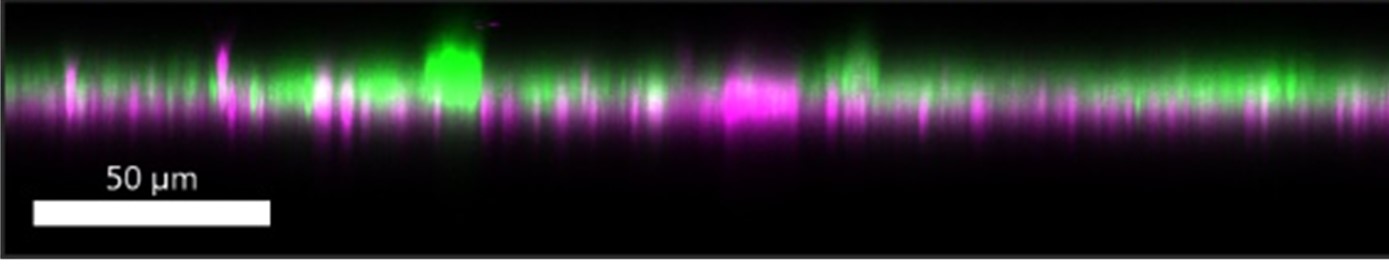
| Fig. 6: ARPE-19 polarization in oRBB-chip devices. Left: Orthogonal views of confocal images taken of ARPE-19 cultures in oRBB-chip devices show apical localization of Ezrin relative to nuclei (blue), ZO-1 (green), and Collagen IV (magenta). Right: Hoechst, and ZO-1 are removed, and the Ezrin channel has been adjusted to green to highlight the apical polarization of Ezrin and basal localization of Collagen IV. Bottom: magnified view of the orthogonal angle. Scale bars: 50 μm. | |
Discussion and Next Steps
For ARPE-19 cultured in oRBB-chip devices, TEER was higher than expected, and small molecule permeability was lower than expected, indicating that the oRBB-chip is able to support the development of functionally tight epithelial barriers. Immunohistochemistry and confocal microscopy confirmed that oRBB-chip ARPE-19 cultures not only had higher expression of key ARPE-19 maturation markers, but were also able to develop cobblestone tight packing and apicobasal polarity. These data also validate the ARPE-19 differentiation protocol established by Hazim et al., which highlights the importance of Nicotinamide supplementation after a 7 day growth period. Based on functional, and morphological metrics, we have established a protocol for reliably developing mature, functionally tight, and highly polarized ARPE-19 barrier tissues in oRBB-chip devices.
This work, which establishes culture and functional assessment methods for RPE barrier tissues in oRBB-chip devices, will be leveraged as a part of the greater effort to develop a complete model of the outer retinal blood barrier. Future analyses may include ARPE-19/HUVEC co-cultures in oRBB-chip devices in order to study the potential effects of endothelial signaling cues on ARPE-19 development, the use of flow modules to study how basal flow affects oRBB tissue development, and the assessment of apicobasal marker expression, including pigment epithelium derived factor and vascular endothelial growth factor.
Acknowledgements:
Advisors: Danielle Benoit, Ruchira Singh, James McGrath
Collaborators/Experimental Contributors: Jordan Jones, Annika Deans, Arvind Srivatsava, Vinay Abhyankar, Mehran Mansouri, Meng Hsu
References:
[1] M. Arya, A. S. Sabrosa, J. S. Duker, and N. K. Waheed, “Choriocapillaris changes in dry age-related macular degeneration and geographic atrophy: a review,” Eye Vis (Lond), vol. 5, 2018/09/15/ 2018, doi: 10.1186/s40662-018-0118-x.
[2] I. Bhutto and G. Lutty, “Understanding age-related macular degeneration (AMD): Relationships between the photoreceptor/retinal pigment epithelium/Bruch’s membrane/choriocapillaris complex,” Mol Aspects Med, vol. 33, no. 4, pp. 295-317, 2012/08// 2012, doi: 10.1016/j.mam.2012.04.005.
[3] A. A. Hussain, C. Starita, A. Hodgetts, and J. Marshall, “Macromolecular diffusion characteristics of ageing human Bruch’s membrane: implications for age-related macular degeneration (AMD),” (in eng), Exp Eye Res, vol. 90, no. 6, pp. 703-10, Jun 2010, doi: 10.1016/j.exer.2010.02.013.
[4] A. D. Kulkarni and B. D. Kuppermann, “Wet age-related macular degeneration,” (in en), Advanced Drug Delivery Reviews, vol. 57, no. 14, pp. 1994-2009, 2005/12/13/ 2005, doi: 10.1016/j.addr.2005.09.003.
[5] W. L. Wong et al., “Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis,” (in eng), Lancet Glob Health, vol. 2, no. 2, pp. e106-116, 2014/02// 2014, doi: 10.1016/S2214-109X(13)70145-1.
[6] K. L. Pennington and M. M. DeAngelis, “Epidemiology of age-related macular degeneration (AMD): associations with cardiovascular disease phenotypes and lipid factors,” (in eng), Eye Vis (Lond), vol. 3, p. 34, 2016, doi: 10.1186/s40662-016-0063-5.
[7] D. Pascolini and S. P. Mariotti, “Global estimates of visual impairment: 2010,” British Journal of Ophthalmology, vol. 96, no. 5, p. 614, 2012, doi: 10.1136/bjophthalmol-2011-300539.
[8] M. J. Ammar, J. Hsu, A. Chiang, A. C. Ho, and C. D. Regillo, “Age-related macular degeneration therapy: a review,” (in eng), Curr Opin Ophthalmol, vol. 31, no. 3, pp. 215-221, May 2020, doi: 10.1097/icu.0000000000000657.
[9] A. Biesemeier, T. Taubitz, S. Julien, E. Yoeruek, and U. Schraermeyer, “Choriocapillaris breakdown precedes retinal degeneration in age-related macular degeneration,” (in en), Neurobiology of Aging, vol. 35, no. 11, pp. 2562-2573, 2014/11/01/ 2014, doi: 10.1016/j.neurobiolaging.2014.05.003.
[10] J. Ambati and B. J. Fowler, “Mechanisms of age-related macular degeneration,” (in eng), Neuron, vol. 75, no. 1, pp. 26-39, Jul 12 2012, doi: 10.1016/j.neuron.2012.06.018.
[11] D. L. Forest, L. V. Johnson, and D. O. Clegg, “Cellular models and therapies for age-related macular degeneration,” Dis Model Mech, vol. 8, no. 5, pp. 421-427, 2015/05/01/ 2015, doi: 10.1242/dmm.017236.
[12] S. Volland, J. Esteve-Rudd, J. Hoo, C. Yee, and D. S. Williams, “A comparison of some organizational characteristics of the mouse central retina and the human macula,” (in eng), PLOS ONE, vol. 10, no. 4, p. e0125631, 2015, doi: 10.1371/journal.pone.0125631.
[13] S. Picaud, D. Dalkara, K. Marazova, O. Goureau, B. Roska, and J.-A. Sahel, “The primate model for understanding and restoring vision,” (in en), PNAS, vol. 116, no. 52, pp. 26280-26287, 2019/12/26/ 2019, doi: 10.1073/pnas.1902292116.
[14] M. E. Pennesi, M. Neuringer, and R. J. Courtney, “Animal models of age related macular degeneration,” Mol Aspects Med, vol. 33, no. 4, pp. 487-509, 2012/08// 2012, doi: 10.1016/j.mam.2012.06.003.
[15] C. J. Zeiss, “REVIEW PAPER: Animals as Models of Age-Related Macular Degeneration: An Imperfect Measure of the Truth,” Veterinary Pathology, vol. 47, no. 3, pp. 396-413, 2010/05/01 2010, doi: 10.1177/0300985809359598.
[16] M. Shah, S. Cabrera-Ghayouri, L. A. Christie, K. S. Held, and V. Viswanath, “Translational Preclinical Pharmacologic Disease Models for Ophthalmic Drug Development,” (in eng), Pharm Res, vol. 36, no. 4, p. 58, Feb 25 2019, doi: 10.1007/s11095-019-2588-5.
[17] M. Mansouri et al., “The Modular µSiM Reconfigured: Integration of Microfluidic Capabilities to Study In Vitro Barrier Tissue Models under Flow,” (in eng), Adv Healthc Mater, vol. 11, no. 21, p. e2200802, Nov 2022, doi: 10.1002/adhm.202200802.
[18] M. C. McCloskey et al., “The Modular µSiM: A Mass Produced, Rapidly Assembled, and Reconfigurable Platform for the Study of Barrier Tissue Models In Vitro,” (in eng), Adv Healthc Mater, vol. 11, no. 18, p. e2200804, Sep 2022, doi: 10.1002/adhm.202200804.
[19] K. V. Manian et al., “3D iPSC modeling of the retinal pigment epithelium-choriocapillaris complex identifies factors involved in the pathology of macular degeneration,” Cell Stem Cell, Mar 29 2021, doi: 10.1016/j.stem.2021.02.006.
[20] Y. T. Tsai et al., “Impaired cholesterol efflux in retinal pigment epithelium of individuals with juvenile macular degeneration,” (in eng), Am J Hum Genet, vol. 108, no. 5, pp. 903-918, May 6 2021, doi: 10.1016/j.ajhg.2021.04.006.
[21] A. P. Voigt et al., “Bulk and single-cell gene expression analyses reveal aging human choriocapillaris has pro-inflammatory phenotype,” (in eng), Microvasc Res, vol. 131, p. 104031, Sep 2020, doi: 10.1016/j.mvr.2020.104031.
[22] T. Kurihara et al., “Hypoxia-induced metabolic stress in retinal pigment epithelial cells is sufficient to induce photoreceptor degeneration,” eLife, vol. 5, p. e14319, 2016/03/15 2016, doi: 10.7554/eLife.14319.
[23] R. A. Hazim, S. Volland, A. Yen, B. L. Burgess, and D. S. Williams, “Rapid differentiation of the human RPE cell line, ARPE-19, induced by nicotinamide,” (in eng), Exp Eye Res, vol. 179, pp. 18-24, Feb 2019, doi: 10.1016/j.exer.2018.10.009.
[24] L. J. Rizzolo, S. Peng, Y. Luo, and W. Xiao, “Integration of tight junctions and claudins with the barrier functions of the retinal pigment epithelium,” Prog Retin Eye Res, vol. 30, no. 5, pp. 296-323, 2011/09/01/ 2011, doi: https://doi.org/10.1016/j.preteyeres.2011.06.002.
[25] Z. Ablonczy et al., “Human retinal pigment epithelium cells as functional models for the RPE in vivo,” (in eng), Invest Ophthalmol Vis Sci, vol. 52, no. 12, pp. 8614-20, Nov 4 2011, doi: 10.1167/iovs.11-8021.
[26] N. M. Korthagen, J. Bastiaans, J. C. van Meurs, K. van Bilsen, P. M. van Hagen, and W. A. Dik, “Chloroquine and Hydroxychloroquine Increase Retinal Pigment Epithelial Layer Permeability,” (in eng), J Biochem Mol Toxicol, vol. 29, no. 7, pp. 299-304, 2015/07// 2015, doi: 10.1002/jbt.21696.
[27] A. Ahmado et al., “Induction of differentiation by pyruvate and DMEM in the human retinal pigment epithelium cell line ARPE-19,” (in eng), Invest Ophthalmol Vis Sci, vol. 52, no. 10, pp. 7148-59, Sep 9 2011, doi: 10.1167/iovs.10-6374.
[28] K. C. Dunn, A. E. Aotaki-Keen, F. R. Putkey, and L. M. Hjelmeland, “ARPE-19, a human retinal pigment epithelial cell line with differentiated properties,” (in eng), Exp Eye Res, vol. 62, no. 2, pp. 155-69, Feb 1996, doi: 10.1006/exer.1996.0020.
[29] R. Toda, K. Kawazu, M. Oyabu, T. Miyazaki, and Y. Kiuchi, “Comparison of drug permeabilities across the blood-retinal barrier, blood-aqueous humor barrier, and blood-brain barrier,” (in eng), J Pharm Sci, vol. 100, no. 9, pp. 3904-11, Sep 2011, doi: 10.1002/jps.22610.
[30] H. Bao et al., “The Interplay Between E-Cadherin, Connexin 43, and Zona Occludens 1 in Retinal Pigment Epithelial Cells,” Invest. Ophthalmol. Vis. Sci., vol. 60, no. 15, pp. 5104-5111, 2019, doi: 10.1167/iovs.19-27768.
[31] T. Kivelä, J. Jääskeläinen, A. Vaheri, and O. Carpén, “Ezrin, a membrane-organizing protein, as a polarization marker of the retinal pigment epithelium in vertebrates,” (in eng), Cell Tissue Res, vol. 301, no. 2, pp. 217-23, Aug 2000, doi: 10.1007/s004410000225.
[32] R. H. Croze et al., “ROCK Inhibition Extends Passage of Pluripotent Stem Cell-Derived Retinal Pigmented Epithelium,” (in eng), Stem Cells Transl Med, vol. 3, no. 9, pp. 1066-1078, 2014/09// 2014, doi: 10.5966/sctm.2014-0079.