Permeability Continued
This is a followup to my previous post, found here. Some of the data in that post is now outdated and will be replaced with newer data here.
I’ve added to the comparison the diffusion of cytochrome C and urea through a polyethersulfone “high-flux” membrane. This material is commercially available just like the cellulose triacetate. In spite of increased transport of cytochrome C, the material’s permeability to urea is actually reduced by comparison to cellulose.
Note that these plots contain slightly different values and error bars than my previous plots. This is due to the other work I’ve done since my last post, concerning quantification and propagation of time error. By measuring the diffusion time for each individual system, I’ve measured the average time for each high-flux experiment and carried the error through to the permeability value. I also retroactively added a corrective factor to the time measurements for my previous experiments, assuming that roughly the same amount of error has been present throughout.
This has had the pleasant effect of making my urea data for NPN appear sane. The permeability of urea through NPN is not significantly different from its permeability through water. The permeability of cytochrome C through the two commercial membranes isn’t significant at alpha = 5%, but is significant at alpha = 10%, so do with that what you will. It’s not clear to me why there was so much error in the cytochrome C high-flux experiments (n = 3), so I may go back and repeat them in hopes of tidying that up. Otherwise, however, this data looks good and confirms our suspicions that our membranes certainly have something to contribute to equilibrium dialysis!
EDIT: According to Clark, et al [1], it seems that the water flux (the value to which the name “high-flux” refers) is not well correlated with solute flux for small solutes such as urea:
A recent study published by Murthy et al clearly illustrates the concept that a membrane’s small solute removal capabilities are not necessarily tied to its water permeability35. Six chronic HD patients were treated with a large surface area unsubstituted cellulosic dialyzer (2.2 m2; KUF, 6 ml/hr/mm Hg) and a high-flux polysulfone dialyzer (1.8 m2; KUF, 52 ml/hr/mm Hg) in a crossover manner. The mean first-use urea clearance was higher, although not significantly, for the cellulosic dialyzer at a blood flow rate of both 300 ml/min (246
6 vs. 241
2 ml/min) and 400 ml/min (288
8 vs. 280
4 ml/min). This equivalence was achieved despite the substantially greater pore size and higher water flux of the polysulfone membrane. Although the greater surface area of the cellulosic dialyzer contributed to this equivalence, the same type of cellulosic dialyzer with a comparable surface area (1.7 m2) has an in vitro urea mass transfer coefficient (permeability-area product: KoA) that is substantially greater than that of the 1.8 m2 polysulfone dialyzer studied (1030 and 945 ml/min, respectively)10. These findings suggest the significantly lower thickness (9
m) of the cellulosic dialyzer relative to that of the polysulfone dialyzer (40
m) was an important factor in this study.
Additional recent data emphasizing the importance of membrane thickness in small solute removal were published by Leypoldt et al40, who determined in vitrourea KoA values for highly permeable cellulose triacetate (1.9 m2; KUF, 36 ml/hr/mm Hg) and polysulfone (1.8 m2; KUF, 55 ml/hr/mm Hg). For blood flow rates ranging between 300 and 450 ml/min and a dialysate flow rate of 500 ml/min, the mean urea KoA value for the cellulose triacetate dialyzer (1070 ml/min) was approximately 43% higher than that of the polysulfone dialyzer (750 ml/min). An increase in the dialysate flow rate to 800 ml/min resulted in a 15% increase in urea KoA for both dialyzers, preserving the difference between the dialyzers on a percentage basis. For these dialyzers of comparable water flux, the probable explanation for this difference was, again, the large difference in membrane thickness (15 vs. 40
m, cellulose triacetate vs. polysulfone).
This paper explains the mechanics of hemodialysis with regard to membrane properties very well, and emphasizes membrane thickness as a key factor — it’s no surprise, then, that our NPN solute flux was so much higher.
[1]: Clark, William R; Hamburger, Richard J; Lysaght, Michael, J. “Effect of membrane composition and structure on solute removal and biocompatibility in hemodialysis”. Kidney International (1999) 56, 2005–2015.
Available: http://www.nature.com/ki/journal/v56/n6/full/4491140a.html
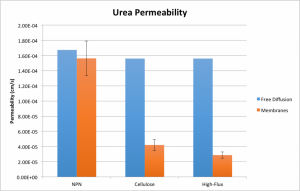
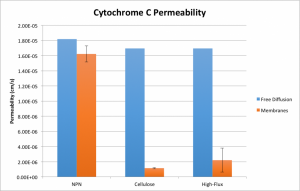
Unless something is mislabeled, the data actually shows the high flux material passing urea slower and cytochrome C faster than the low flux membrane material.
Whoops, guess I unknowingly gave into some confirmation bias here. I’ve corrected the post and will investigate the material’s supposed properties tomorrow, to make sure this makes sense…