μSiM Vasc-SkM update 2024.03.13
The μSiM Vasc-SkM Microphysiological development can be separated into 4 parts:
- Media tuning for integrated culture
- Force sensing element setup
- Assembly of the μSiM Vasc-SkM and setting up the perfusion
- Transendothelial Electrical Resistance (TEER) measurement of the Vast component
Work has been done on the first 2 parts.
Media tuning for integrated culture
In order to find a media recipe compatible with the integrated culture of the blood vessel mimic and myobundle, various media compositions and additives have been tested in 2D and 3D culture. We have found 2 promising candidate media conditions for culturing the blood vessel mimic and myobundles together (Diff:EC2 75:25 media + VEGF and Diff:viSMC 75:25 media + VEGF).
Cord blood derived endothelial cells (CBEPC)
We first tested different media recipe in the CBEPCs cultured in 2D plates due to their ease to culture. The CBEPCs were grew to confluent, and then cultured in the different test media for 4 days, with phase contrast images taken (Figure 1). Below are the media compositions tested without or with 10ng/mL vascular endothelial growth factor (VEGF):
| Media recipe tested in CBEPC: |
|
We found that Diff:EC 9:1 and Diff:EC2 75:25 media caused reduced confluency after 4 days, but adding VEGF to the media improved EC morphology and coverage, leading to similar confluency and morphology compared to the control case cultured in EC media. However, the Diff media was too detrimental to the CBEPCs, and adding VEGF could not save it.

Figure 1. Phase contrast image of CBEPC before (top row) and after 4 days (bottom row) of culture in different media.
Human skeletal muscle cells (hSkM)
Based on the result from CBEPC plate culture, we then tested the following media for 4 days in healthy hSkM cultured in 2D:
| Media recipe tested in healthy hSkM: |
|
Just by looking at the phase contrast images of the healthy hSkM before and after their culture in the different media, the effect of different media could not be distinguished. So, we then used the media compositions 1, 3, 4, 5, 6 in the 2D hSkM culture, and tested them in myobundles made with healthy myoblasts (Figure 2). The myobundles were fabricated and cultured in growth media for 4 days, and then they were shifted into Diff media for 7 days before they were cultured in the different media compositions for 4 days prior to force test. Healthy myobundles cultured in Diff:EC 75:25 + VEGF and Diff:EC 9:1 did not have significantly different contractile profile compared to those cultured in Diff media. Hence these two media will be used in following experiments. In addition, due to the need for switching from the carbon electrodes to platinum (Pt) electrodes for better compatibility with the μSiM Vasc-SkM during force test, we force tested myobundles cultured in Diff media for 11 days total with carbon electrodes and Pt electrodes. The contractile profile of myobundles was not significantly different between those stimulated by Pt electrodes and those stimulated by carbon electrodes.
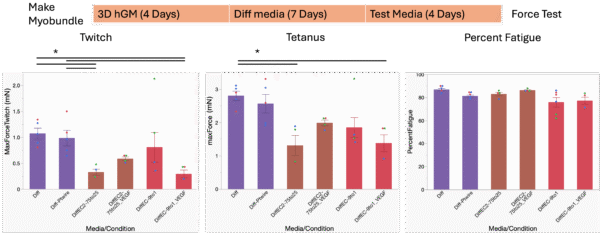
Figure 2. Culture timeline and contractile profile of healthy myobundles with Twitch (1 stimulation, 1Hz), Tetanus (20 stimulation, 20Hz), and Percent Fatigue (percent force reduction after 30s of 20Hz stimulation). Myobundles in Diff-Ptwire group were stimulated by Pt electrodes. Myobundles in all other groups were stimulated by carbon electrodes as usual. Mean±SEM, N = 3-6 myobundles per treatment. One-way ANOVA, *P<0.05.
As we will use the μSiM Vasc-SkM for the study of rheumatoid arthritis (RA) and atherosclerosis, and myobundles made with RA myoblasts are more sensitive to undesirable culture conditions, we tested the media compositions in RA myobundles as well. 75% Diff media with 25% viSMC culture media (Diff:viSMC 75:25) with VEGF was also tested as one potential media composition. Although all three media compositions (Diff:EC2 75:25 + VEGF; Diff:EC 9:1; Diff:viSMC 75:25 + VEGF) caused significant force reduction in RA myobundles compared to the Diff media, the reduction caused by Diff:EC2 75:25 + VEGF and Diff:viSMC 75:25 + VEGF was smaller and bearable (Figure 3). Hence, Diff:EC2 75:25 + VEGF and Diff:viSMC 75:25 + VEGF will be tested further in viSMC and viEC.
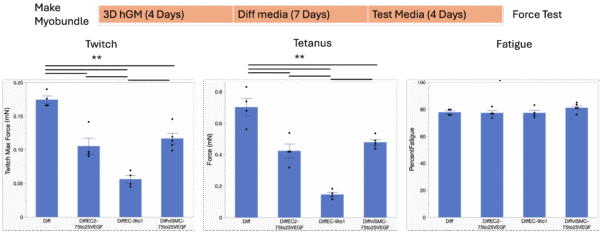
Figure 3. Culture timeline and contractile profile of RA myobundles. Mean±SEM, N = 4-5 myobundles per treatment. One-way ANOVA, **P<0.01.
iPSC derived smooth muscle cells (viSMC)
viSMC was cultured in 2D to confluent and then for 4 days in Diff media, Diff:EC2 75:25 + VEGF and Diff:viSMC 75:25 + VEGF, with phase contrast images taken before and after the 4 days of media composition test (Figure 4). Just by observing the phase contrast image, we did not find distinct difference between the three media used. Hence immunostaining for viSMC maturation will be done.

Figure 4. Phase contrast image of viSMC before (top row) and after 4 days (bottom row) of culture in different media.
Force sensing element setup
The 3 main parts of the force sensing element include making flexible frames, i.e. PDMS frames, culturing myobundles on PDMS frames, and analyzing the force of myobundles on the PDMS frames.
PDMS frame fabrication and calibration
3D printed master molds were used to make crude PDMS frames to test the concept (Figure 5A). We hooked paperclips to the beams on the PDMS frames and measured the deflection of the beam as it was pulled by the weight of the paperclips (Figure 5B). The weight of the paperclips was converted to the gravity force applied on the beams, plotted with the length of deflection of the beam, and fitted with a best-fit-line for the calibration lines (Figure 5C). Nicely fitted calibration lines with R2 values ranging from 0.97-0.98 were obtained for the beam in each frame, but the slope of the calibration lines was not very consistent across the frames. This varying slope could be caused by batch variability in PDMS curing temperature and inconsistency in frame thickness when they were made with the master molds. As the current master mold could not completely replicate the design of the PDMS frame, and could not control frame thickness nicely either, we are making molds for die casting the PDMS frames. Better oven with more precise temperature control will also be used to reduce batch variability for cured PDMS stiffness.
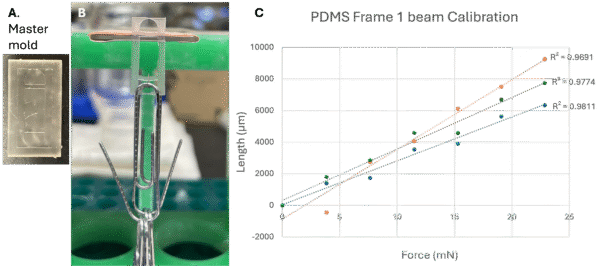
Figure 5. 3D printed master mold for crude PDMS frame making, PDMS frame calibration with paperclip, and the calibration curves for length of beam deflection v.s. force applied on beam by paperclip weight.
Myobundle culture on PDMS frames
Then we made molds that will enable us to make myobundles attached onto the beams of the PDMS frames (Figure 6B). We cultured the myobundles attached to PDMS frames for 4 days in growth media, and 11 days in Diff media (Figure 6A). Over the 11 days of culture, the myobundles gradually detached from the PDMS beams (Figure 6C).
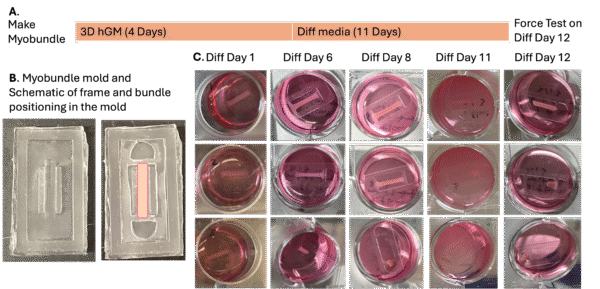
Figure 6. Making and culturing myobundles attached onto PDMS frames.
Force testing myobundles on PDMS frames
Though most of the myobundles detached from the PDMS frames before force testing on Day 12 of differentiation, we were able to take a video of the myobundle that survived. Pt electrodes that were used here to stimulate the myobundle were found capable of inducing comparable contraction to carbon electrodes that we usually use for force tests (Figure 2). The plate holding myobundle was set on a heater to ensure that the myobundles were in 37˚C media. In the video for twitch, the myobundle was able to pull the PDMS frame as it was stimulated. In the video for tetanus, the myobundle pulled itself off the PDMS frame mid-way through the continuous stimulation. As the myobundles seem to have trouble attaching to the PDMS frames tightly enough during culture and force testing, we will try pre-coating the PDMS frames with polydopamine (PDA) to improve the hydrophilicity to allow better gripping of the myobundles to the beams.