R/C TEM scalebar calibration
Last week, I took some of Jess’ 20 nm Au colloids and placed them on a membrane for imaging under TEM. I took images at each of our standard magnifications. Using Mike B.’s pore counting program, I generated statistics and calculated the pixel-to-nm conversion ratio assuming the gold colloids were 20 nm (< 10% variation).
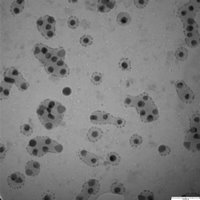 Here are the numbers:
Here are the numbers:
35 kx – 1.21 px / nm
50 kx – 1.54 px / nm
75 kx – 2.26 px / nm
100 kx – 3.29 px / nm
150 kx – 5.28 px / nm
This confirms that the 75kx and 100kx setting on the digital camera software was incorrectly entered. I’ll notify Brian asap.
From data obtained today, I’m hesitant to calibrate the TEM using gold particles. The Malvern measures the 20nm gold as 24.9nm in diH2O and 15.3nm in 10mM HEPES. We wanted to then use the TEM to understand the results from the Malvern sizer. Course calibrating the TEM to these standards presents a bit of a problem.
Just out of curiosity, is there some sort of “gold” standard (pun not really intended) that TEM can be calibrated with? Like something based on atomic distances perhaps?
Something else I just thought of:
What membrane is this and why aren’t there any larger pores? Is this a central sample? I’m trying to set up an experiment right now to get the gold to diffuse through a membrane, and I don’t think the one in this picture would be of any use.
Hmm…The variability of these colloids looks a lot larger than 20%. I wonder if this is a bad batch.
I spoke with Karen on Monday about calibration, and since her TEM is used for Pathology, it is calibrated regularly, so we can rely on the scale bar that she gives us. She has some kind of calibration reference, but she was a couldn’t think of what it was made of. If we find out what she uses, we should be able to calibrate Brian’s TEM.
Ok, I’ll check with Karen about the standards.
The membrane I used was from the center of wafer 304.