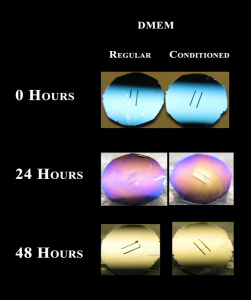
Conditioned DMEM – Discoloration
Motivated by Tom’s suggestion at lab meeting on Tuesday, I tried “conditioning” the DMEM+10%FBS before adding the chips. “Conditioning” just means that I allowed the DMEM+10%FBS to equilibrate in the incubator for an hour before adding the chips to test if a brief (~1 hour) exposure to very high pH causes discoloration. DMEM+10%FBS without the 1 hour incubation was the control.
Unfortunately, conditioning the media did not seem to inhibit discoloration since both chips discolored at the same rate. I’ll repeat next week.
Please add a few more details on the rationale behind this experiment for those of us who were not at the meeting. What is the initial pH for non-conditioned media? Do we know it takes ~ 1 hr to reach equilibrium?
I wrote this as a comment elsewhere, but I’ll repeat it here in case it was missed. I think we should test if membranes discolor in MES buffer vs. TRIS buffer each titrated to their pKa. One is slightly basic (<6.5), one is slightly acidic (>8). If discoloration is strictly pH then the TRIS buffer should discolor in a day as it is even more basic than DMEM in the incubator. We tried these experiments way back and did not see any discoloration, but we did not put the mixtures in the incubator. If we do this and see no discoloration we should also try TRIS buffer plus salts. I’ve never been sold on the pH only explanation of this phenomenon, but I might buy a pH + salts explanation.