Fluorescent HUVECs in PC transwells
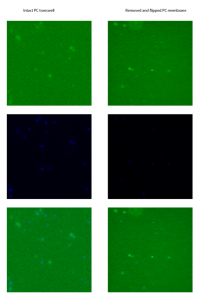
I wanted to do some more fluroescent imaging in transwells to look into background fluroescence. This was mainly motivated by my inital study. These are HUVECs stained with 5uM CMFDA and 10uM Hoechst 33342, fixed in formaldehyde and imaged on the Zeiss at 10X. This was 6 hours after seeding. Before acquiring the images, I did autoscale in TCGui.
I read some papers that reported cutting membranes out of transwells in order to do microscopy. I tried this to see if it made a difference (cut and flipped membranes were aligned so that the cells faced the objective). The top row is CMFDA stained cells, the middle row is Hoechst and the bottom row is the overlay. All post-acquisition processing was in ImageJ.
First, I think the CMFDA was a bit old, so the staining was pretty faint. Autoscale increased the background quite a bit. In the normal transwell setup (imaging through the intact transwell), the cells appeared as blurry blobs – no resolution. Cutting out the membrane and flipping it increased the resolution dramatically. No wonder people do it – but it’s not ideal. Unfortunately the Sepcon pnc-Si sample that I wanted to image broke. I think a side by side comparison between PC transwells and pnc-Si will be pretty dramatic. Hopefully I’ll get this next week.
I used CMFDA in my cell culture experiments and I reckon that it didn’t work well with HUVECs. But with fibroblasts the staining was much better. Also I avoided autoscaling in TCGui and got clear background. Did you use CMFDA and Hoechst at the same time?
I think maybe the CMFDA was just too old. I got brighter staining the 1st time I used it. I added the Hoechst after the CMFDA incubation.
Can you please upload the protocol for Hoechst staining.