Long-term viability – pnc-Si and PET transwells
Some transwell cell culture models require 1 week or more of cell growth in order to let the cells fully differentiate and form mature monolayer barriers. I had a couple of leftover samples a couple of weeks ago, so I measured the viability of cells on RTP’ed pnc-Si and PET transwells 7 days after plating the cells. These were b.End3 cells (P9) stained with the Live/Dead assay kit from Invitrogen. The images were pseudo-colored in ImageJ but not adjusted for brightness/contrast.
Here is the pnc-Si transwell. One membrane window was completely broken (the right/top) and the other one had a small tear. Although the cells were largely confluent, there were quite a few dead cells. The dead cells seem to be concentrated in a pattern emanating from the broken area of the left/bottom membrane and close to the other window. It’s possible that a lot of cells died when these membranes broke (probably during media changes), perhaps due to a burst of fluid flow through the window area?.
This is an image taken “far” from the membrane slits of a pnc-Si transwell. The Live/Dead ratio is drastically different from the above image. The cells are confluent, nicely stained and largely alive. The dead cells are concentrated at the top-left of the image – near the O-ring of the housing. Perhaps there is an edge effect at this interface?
Here is the dead stain channel from a difference region of interest – the membrane windows are totally black. The orientation is the same as the top image – the right membrane is completely broken but the left membrane is intact. The dead cells seem to be concentrated at the interface of the free-standing and supported membrane areas – and especially around the broken membrane. Taken together, these images suggest that if membranes break during the culture period, cells are adversely affected near the membranes but not far from the membranes. This might be due to shearing forces by fluid flow through the open slits. It’s also possible that ECM-mediated or intercellular signals propagating from the broken membrane (a monolayer ‘wound’) could cause some of the cells to die.
By contrast, there are very few dead cells on PET transwells (this is a live/dead overlay):
I counted the live and dead cells with the MATLAB cell counter and then calculated the % viability. These data were collected from 2 different images:
Viability is essentially 100% on PET transwells, but only ~ 80% on pnc-Si transwells. For pnc-Si, the viability was only ~65% near the membrane but ~94% far from the membrane, which caused the rather large deviation in the pnc-Si data.
The pnc-Si samples were not discolored when I did this experiment, and you can see from the images that the membranes are stable out to a week in the presence on cells. It looks like cell viabilities on pnc-Si will closely match those of PET if the membranes remain intact for the entire culture period. Even with membrane breakage, ~80% viability on pnc-Si isn’t TOO bad.



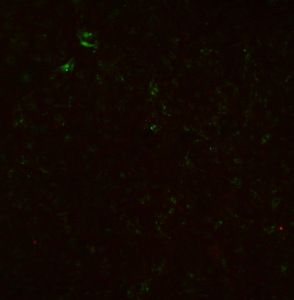

Are the cells imaged through the membrane in the PET transwells or are the cells plated on the membrane facing down? Why is the stain so dull, unlike pnc-Si?
In this 7day period, were the cells in upside down configuration? How long after the adhesion period were the inserts put back into the plate wells?
Cells plated on the underside of PET transwells. Ditto for the pnc-Si transwells. I did not adjust the contrast/brightness on these images, so the green is kind of faint. Even so, the staining looks different – I’m not sure why.
I allowed the cells to adhere for 4-5 hours and then I flipped the samples into 24-well plates. So the cells were growing “upside-down” the entire 7 day timeframe.