Photobleaching
The mechanism of organic dye photobleaching is not well understood, but there are a few methods out there for reducing the rate of photobleaching. Some literature suggests that photobleaching is due to excess oxygen in solution and the interaction between the dye and oxygen radicals. Actin research swears by the following “anti-bleaching buffer” to limit the oxygen in solution (Harada et al. 1990 JMB):
- Glucose
- Glucose Oxidase-FAD
- Catalase
- Beta-mercaptoethanol (BME)
Glucose oxidase-FAD oxidizes glucose to gluconolactone, and in the process of regenerating its attached FAD moiety, creates H2O2. Catalase breaks down two H2O2 into 2 H2O and 1 O2. One time around this chemical cycle will use 1 molecule of O2 and produce 1/2 molecule, leading to the overall removal of oxygen.
The BME is a reducing agent (or antioxidant) that is added to the mixture as it seems to offer additional protection to the fluorophore. Harada et al found that while BME does reduce photobleaching alone, the combination of BME with the oxygen scavenging system is much better.
A recent paper (van Dijk et al. J Phys Chem B 2004) concerned with photobleaching of fluorophores in optical traps looked back at the “anti-bleaching buffer” and compared it to deoxygenated systems and the use of the antioxidant ascorbic acid (Vitamin C). The results from this paper that deoxygenated systems had no effect on photobleaching, while both “anti-bleaching buffer” and ascorbic acid reduced bleaching. Ascorbic acid worked much better, and the authors suggested that the only reason that “anti-bleaching buffer” worked in the first place was because it also had the antioxidant BME (which conflicts a bit with the Harada findings).
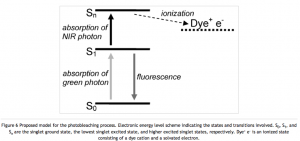
The van Dijk paper suggests that a fluorophore bleaches because it attains a higher excited state that can undergo ionization, which kills the fluorescence properties of the molecule. See the following figure:
Antioxidants could fix the fluorophore by reducing it, thereby returning an electron and allowing the molecule to remain in its neutral fluorescing state. BME is an antioxidant, but it can also reduce disulfide bonds. Therefore it should only be used in conjugation with proteins found in the cytosol (like actin). The cytosol is a reducing environment and no proteins (except for environment monitoring alarm proteins) have disulfide bonds. Proteins found on the cell surface (like integrins) or excreted proteins (like albumin) contain many disulfide bonds and should probably be used with another antioxidant like ascorbic acid.
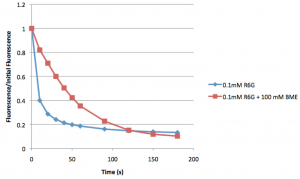
I did a couple of quick tests with 0.1 mM free rhodamine 6g to check these ideas. The first set were performed with a large drop on a slide. The rhodamine excitation beam was left on and images were taken every 10s. Bleaching was quantified using ImageJ. The following figure compares the control to R6G with BME (Horizontal axis is the percentage of initial fluorescence):
You can clearly see there is very little photobleaching with the BME sample. Next I performed the same experiments, but this time with a smaller volume squished between the slide and a coverslip:
This time you can see that both samples bleach, but the sample with BME bleaches much slower than the control.
These simple experiments show that just by adding some BME to a sample (as long as it includes only cytosolic proteins) can reduce bleaching. It would be interesting to compare this to BME + oxygen scavengers or to ascorbic acid.

I’m pretty surprised that the mechanism is still controversial. What is being reduced when Glucose is being oxidized in the antibleaching buffer? Why isn’t this combination considered a ‘reducing agent.’
When glucose is oxidized, the FAD complexed with the glucose oxidase is reduced to FADH2. It is then regenerated into FAD with the addition of oxygen and subsequent formation of H2O2. If this is how the cycle proceeds every time, it doesn’t seem like there’s any reduction of the fluorophore. But maybe instead of the oxygen being the electron donor for the reduction step, the oxidized fluorophore is used.
100mM is a lot of BME, but I gave that a shot because that’s how much ascorbic acid was being used. I didn’t check the pH though. That’s a good point, not only for the proteins but also for the fluorophores.
Sorry wrote that a little backwards. The fluorophore may accept the electron during the reduction step instead of FAD. FAD is a cofactor to the enzyme in this reaction, and I’m not sure but it may just diffuse into the active site during the reaction. That being said, given enzyme specificity, I don’t believe that the fluorophore would be reduced in this step instead of FAD.