PDMS-filled wells don't increase sensitivity for HMM detection
This post is related to the HIV drug screening project that Carrie has been working on. To refresh everyone’s memory, the aim of this project is to use pnc-Si as a filter to separate high molecular mass (HMM) protein-RNA complexes from smaller protein monomers/multimers. Small molecules that break down the HMM form of this protein into smaller pieces would be considered a ‘hit’ in a high throughput screening assay.
We’ve tried these experiments with 3×3 pnc-Si grids on 384 well plates. With this set-up, 1-2uL of protein is added to the well side and the grid is placed on a 384-well plates with pre-filled wells of buffer (30ul). Therefore, we are dealing with a 30-fold dilution, assuming complete equilibrium. Since the HMM that Harold provides to us is only 1-2 mg/mL, this 30-fold dilution drives the protein concentration down to undetectable levels (by Bradford). One way to circumvent this problem is to try to decrease the volume of the wells (another way is to use a more sensitive protein assay). I think Carrie has tried this once but found no benefit. This was my attempt:
I filled the wells of a 384-well plates with 2 ‘drops’ of PDMS from a 1mL syringe and let the PDMS cure. This PDMS plug decreased the well volume to ~15-20uL instead of 30uL. I then set up standard curve protein concentrations of BSA (1 mg/mL -> 0) and HMM(2.2 mg/mL -> 0) in both PDMS-filled wells and normal wells. I pre-filled wells with 30uL buffer (PBS for BSA, HMM buffer for HMM), carefully overlaid the 3×3 SC603 grid and added 2uL of protein to the well side. On each grid, I had membrane windows with and without pinholes, so there was a built-in positive control. I placed the 384-well plate in a humidity chamber and let it sit at 4C overnight.
The BSA standard curves were the same in PDMS-filled and empty wells. However, the HMM standard curve in PDMS-filled wells was quite a bit lower than in empty wells. This means that HMM but not BSA was adsorbing to PDMS over the 24-hour timeframe (or I made a mistake – this is only n=1). Notice how much lower the absorbance values were for HMM compared to BSA.
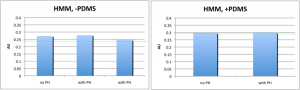
Diffusion (HMM data only):
In empty wells (left panel), there was no difference in the filtrate protein concentration between membranes with and without pinholes (PH). The absorbance values were about the same as the buffer alone (green line). In PDMS-filled wells, the results were the same, although the background absorbance was a bit higher. Therefore, blocking part of the well volume with PDMS in order to decrease the dilution factor by ~2 did not increase the sensitivity of this assay to detect HMM diffusion through pnc-Si.