Diffusion through nanoporous membranes as a function of well height
For a while now I’ve been working on developing and testing the practicality of using nanoporous membranes as filters for highly sensitive assays. Traditional filter-based assays are restricted in their speed by both the time it takes for a particle to find a pore as well as the time it takes for the particle to travel through the pore and come out on the filtrate side of the membrane, but it has been known since the Nature publication (PubMed) that a unique facet of ultrathin membranes is the relative negligibility of the time for diffusion through a pore — that is to say, ultrathin membranes allow molecular separations (of particles of diameter less than roughly a third the pore diameter) to become mostly just a function of particles’ ability to find a pore.
Because of this, lately I’ve been working on a modification to the CytoVu design that would exploit this feature in order to speed up low-volume, low-concentration separations. By reducing the height of the gaskets which form the borders of the filtrate and retentate wells, the particles are more likely to be close to the membrane — and thus, to finding a pore — at any given time, accelerating diffusion.
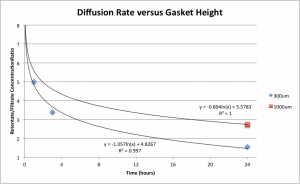
Previous setups employed 1mm (1000um) thick PDMS gaskets on either side of the membrane, so I explored the results of a reduction to 300um:
All collected data are shown — the curves intersect arbitrarily close to t = 0 (since no diffusion has occurred,) so a time of t = 0.01 hours was plugged into the regression curve found for the 300um system and the same point was assumed to be shared by the 1000um system and used for the logarithmic regression, for visualization purposes. Due to the significant time investment each one of these points represents, no more than a single test was performed at any given time, precluding any sort of error bars or standard deviation for a given point, but due to the extremely large (i.e., close to one) and consistent R-squared values achieved on each of the linear regressions of the standard concentration curves used to measure these values, overall error is estimated to be fairly small relative to the scale of the experiment.
These results are promising — no one wants to wait 24 hours for their assay to reach close enough to equilibrium to be conclusive when they can instead wait 5. This also paves the way for even more effective modifications — I’m currently exploring the practicality of employing even thinner gaskets at 100um, which if successful should result in another dramatic reduction in wait time for the system to reach any given ratio of diffusion. Other modifications in the same vein may be worth exploring as well; for instance, increasing the height of the filtrate gasket relative to the height of the retentate gasket would reduce the concentration of particles in the filtrate for any given amount of diffusion, but would also reduce the rate of backward diffusion (from filtrate to retentate) relative to the rate of forward diffusion, reducing the time to equilibrium — such a setup could prove useful in cases where more filtrate volume is required for testing or when a smaller sample volume for the filtrate is available, or if testing the filtrate depends upon the mass of particle rather than the concentration.
UPDATE 5/12/14: I finished the 100um 1 hour test, and I’ve included it in the plot below. Otherwise, this plot represents the same data as before, but with the ratios inverted for easier understanding. On this plot, a ratio of 1 is equilibrium while a ratio of 0 represents no diffusion. A ratio of 0.5 represents twice as much phosphatase in the retentate as is in the filtrate, and so on. The regression method was also changed, but neither this power-law regression nor the former logarithmic regressions should be taken to be meaningful outside the bounds of the observed data, as the diffusion process does not strictly follow such a simple relationship.
This change in diffusion speed for the 100um system is as expected in direction, but not in magnitude. Time to 50% sieving coefficient goes roughly as gasket height squared, so the ratio between the time it takes any two systems to reach a concentration ratio of 0.5 should be equal to the ratio between the squares of the gasket heights; e.g., for the 300um versus 1000um case is, the 300um time to 0.5 is known to be roughly equal to 12 hours, so the time for the 1000um system to reach the same point is equal to roughly 12*((1000^2)/(300^2)) = 133.33 hours. Similarly, for the 100um system, we would expect the time to 0.5 to be roughly 12*((100^2)/(300^2)) = 1.33 hours, but after 1 hour the system is still only about halfway to 0.5 and necessarily slowing down (as the rate of diffusion only decreases as the system approaches equilibrium,) indicating that this prediction is not strictly met by the system.
This is not particularly surprising after having performed the experiment. With such small gaskets (and thus, small volumes, on the range of 4-7 uL) the surface tension of the water starts to have very significant effects on its shape within the wells. Rather than conforming to edges of the PDMS gaskets and filling out the wells, the solution tended to prefer to form droplets, thereby increasing the effective diffusion length significantly beyond the height of the gaskets.
Nevertheless, an increase in diffusion rate above that observed in the 300um system was observed. I don’t personally feel that any further reduction in gasket height is particularly practical due to assembly constraints (as it is, 100um PDMS is very difficult to work with) and the surface tension. Any further gasket improvements will likely have to come in the form of different gasket shape, and/or the relationship between the heights of the two gaskets as discussed earlier. That said, the current system is highly effective for low concentration assaying, capable of providing high sensitivity over a relatively short time — a concentration ratio of 0.25 is more than enough to confidently measure the output of this system after only a single hour!