Residue on SiO2 membranes is dependent on wafer lot, but not storage location
Last meeting, we discussed the possibility of the storage gel-boxes outgassing a residue onto the porous SiO2 membranes, as evidenced by cells not adhering to the membranes and difficulty in acquiring SEM images.
Possible Gel-Box Outgassing on Porous SiO2 Membranes


Several cleaning methods were utilized in attempt to clean the membranes, such as O2 plasma, Argon plasma, Piranha, and Ethanol + NaOH, but none were successful in improving the imaging quality.
Interestingly, while Aslan was imaging plasma-cleaned samples, he took some very peculiar images of the “clean” control sample, an SiO2 membrane from wafer 1234 that had been stored in a 24-well plate for several months. Please note that this is the sample we have been consistently imaging across all of our cleaning attempts in order to “standardize” the SEM.



In response to this, we decided to image a “dirty” chip from wafer 1234 that had been stored in a gel-box. Please note that until this point, all of our “dirty” chips that we have been attempting to clean have been from wafer 1110.
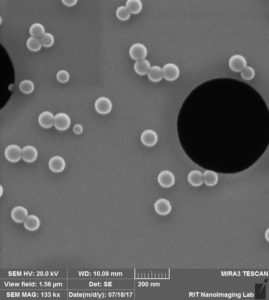
Much to our surprise, the “dirty” chip from wafer 1234 was in fact not visibly dirty. Unlike the “dirty” chips from wafer 1110, there was no visible residue and we could acquire clear SEM images of these chips.



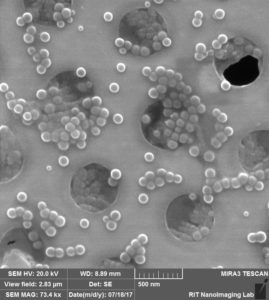
Next, we coated new “dirty” (gel-box stored) and new “clean” (24-well plate stored) chips from wafer 1234 with 50 nm beads. Again, we were able to acquire clear images of the “dirty” chip.
“Dirty” (gel-boxed stored) chip from wafer 1234


“Clean” (24-well plate stored) chip from wafer 12134

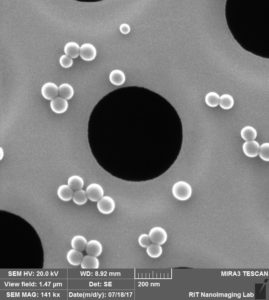
Conclusions:
First, we still do not have a concrete explanation as to why Ana had difficulty seeding cells on the membranes during her focal adhesion experiments in spring 2017. We speculate that the gel-boxes may still be out-gassing onto the membranes despite the clear images we are able to acquire. Tom supposes that the gel-boxes may be out-gassing small, short-chain molecules that are adhering to the surface of the membranes. This prevents proteins from sticking to the membrane surfaces, which negatively impacts cell attachment and spreading. Moving forward, we will only be using chips from wafer 1234 (0.5 um) and 1232 (3.0 um) and all membranes will be oxidized with O2 plasma (2 min, high RF) prior to integration into CytoVu devices. Hopefully, this cleaning step will increase the hydrophilicity of the membrane surface in order to facilitate protein attachment and subsequent cell attachment and spreading.
Second, these experiments have shown that chips originating from the same wafer may not be identical and that not all membranes have through-pores of the appropriate diameters due to errors in the fabrication process, such as under-etching the Si wafer. Moving forward, we will examine the membranes using the 63x objective on the optical microscope in order to ensure that there is no visible pore occlusion on the membranes prior to integration into CytoVu devices.
Third, the image-blurring residue on chips from wafer 1110 is still a mystery. We do not know whether this is fabrication or gel-box related.
Fortunately, we are able to move forward with our adhesion experiments using plasma-cleaned SiO2 membranes from wafers 1234 and 1232. Thank you to Aslan, Marcella, and David for their assistance.
Can you bring a sample or two of each to the next NRG meeting? I’m going to have them tested on the XPS.