Distributions of in-Situ formed Au Nanoparticles
Jirachai’s recent post on the formation of Au nanoparticles on the surface of the NPN is very exciting. For one, it is a very dense monolayer of particles. This self assembly mechanism is magnificent whereas if we tried to dry down a layer of particles, we would get a cake layer similar to what Josh Winans imaged. Crossectional views of the particles show a rough sphericity (larger particles are thicker, smaller particles are difficult to see).


I’m curious about what is causing the nanoparticles to form in the distribution that they are currently made. To my eyes, the largest particles are formed in the regions where the largest distance between pores exists. Smaller particles tend to form in narrow gaps between the pores. So my belief is that the localization of the pores relative to the gold film changes the resultant distribution of formed nanoparticles. Furthermore, I want to know if the nanopores ‘interrupt’ the formation of larger nanoparticles. For comparison’s sake, I’ve found a paper (Gold Nanoparticle Formation Heating) that describes the formation of Au nanoparticles on glass using the PVD/heating method Jirachai used.

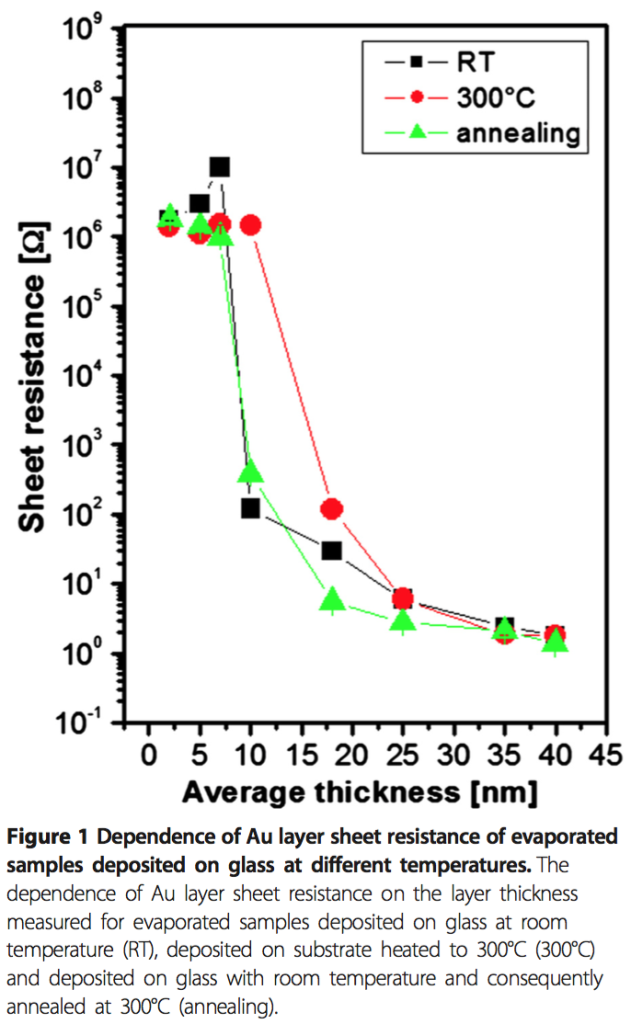
I believe that if we use a substrate with a different distribution of pore sizes and porosity we will be able to change the resultant nanoparticle distribution. If the same pore distribution exists, but has lower porosity, I expect we will have a trend towards larger nanoparticles, fewer overall particles. If the same porosity exists, but a smaller average pore size, I expect a particle distribution with less spread, with more overall particles.
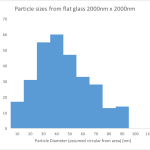
How might we test this hypothesis? We can observe the distributions of formed particles on different substrates. Threshold the images and count particles. From the paper, I created a histogram from their 7 nm gold image (2 micron square):
A simple analysis was done by Kevin on Jirachai’s substrate.
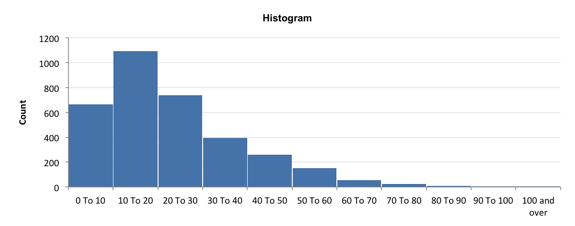

Basically, we are able to discern very small particles in Jirachai’s SEM image vs the AFM image (the total area is similar), so there are many more particles. The particle distribution is still shifted lower on Jirachai’s sample (10 nm, 200 C) vs the paper’s sample (7 nm, 300 C), though my thresholding is debatable. We could try the same process on a nonporous substrate as a control.
We could also localize the specific particles to areas in the SEM image. These localizations could then be correlated with another pattern: the pattern of nanopores. If I am correct, the nearest neighbor distance (NND) will be much smaller between the smaller nanoparticles and the pores than the larger ones. This would be evidence for the idea that the tiny nanoparticles can only form in the restricted spaces, but not very good evidence, because the particles themselves are tiny, which means you can cram more of them into a restricted area. Also, the larger particles are probably absorbing smaller ones. We could also change the deposition film thickness, which would change the surface properties of the gold, influencing the nanoparticle formation (thicker layers have larger nanoparticles in general).
Thoughts or comments are welcome.