Investigating the potential of astrocytes as a neuroinflammation markers by assessing their activation state
Introduction
Astrocytes, which are the most abundant glial cells and key components of the BBB, are known to play crucial roles in initiating and responding to neuroinflammation.1 Astrocytes’ activation after injuries suggests that they could potentially serve as a marker of neuroinflammation. In this blog, I investigated the potential of astrocytes to serve as a reporter of neuroinflammation for in vitro human BBB models. Astrocytes were cultured and exposed to deleterious factors to induce the inflammation and then assessed the expression of glial fibrillary acidic protein (GFAP) and morphological changes characteristic of activation.
Methods
Normal human astrocytes (NHA) were purchased from Lonza Biosciences and used between passages 2-6, following the recommended guidelines. NHAs were seeded at 35,000 cells/cm2 on the 1-slot membrane µSiM precoated with collagen I/fibronectin (100 µg/ml rat tail collagen I and 50 µg/ml of bovine fibronectin), as mentioned in the previous papers.2,3 After reaching confluency within 2-3 days, variable concentrations of cytomix, an equimolar cocktail of tumor necrosis factor-alpha (TNF-α), interferon-gamma (IFN-γ), and interleukin-1 beta (IL-1β), were added to the basal side of the devices for 16 – 20 h to simulate the infiltration of cytokines to the brain (Figure 1). To determine the optimal concentration of cytokines, 10 ng/ml and 100 ng/ml of cytokines were initially tested as reported in other in vitro BBB models.4-6 Phase contrast images of both cytokine-stimulated and non-stimulated astrocytes were captured. Then both groups of astrocytes were stained for GFAP, a marker for astrocyte activation.7,8 Mean fluorescent intensity of GFAP protein per cell, length of astrocyte branches, and branch numbers were calculated using Single cell skeletal analysis on ImageJ9 (Figure 2). By comparing these values between stimulated and non-stimulated astrocytes, I can assess the effects of cytokine exposure on astrocyte activation and morphology.
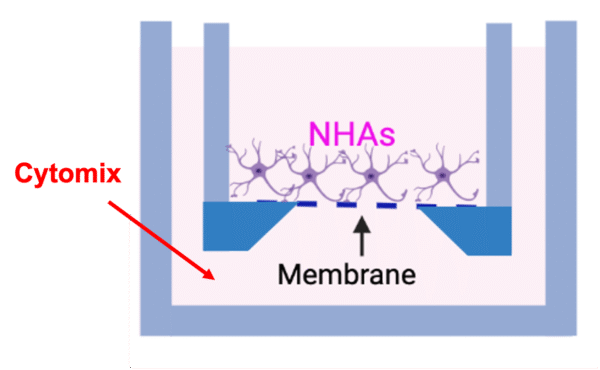
Figure 1 Add cytomix (the analogue of cytokine storm – a cocktail equimolar of TNF-α, IL-1β, and IFN-γ) to the basal compartment of the device to simulate cytokine infiltration to the brain.
Results
My preliminary data demonstrate that cytomix induces a dose-dependent increase in GFAP expression in astrocytes. Immunofluorescence analysis revealed that the expression of GFAP amplified in response to cytomix stimulation, with higher levels observed at higher concentrations (Figure 2B left). Morphological changes were also observed in both phase contrast images and GFAP expression in the cells (Figure 2A). Specifically, simulated astrocytes displayed a more dendritic-like shape, with hypertrophic cells featuring long branches (Figure 2A, blue arrows) and enhanced branch lengths and numbers in response to higher doses of cytomix. Statistical analysis of branch lengths and numbers also confirmed the dramatic increase in cytomix groups compared to the control (astrocyte media). Furthermore, the branch lengths and numbers significantly increased from 10 ng/ml to 100 ng/ml, indicating a potential dose-dependent influence of cytomix on the morphological changes as well. These preliminary findings provide strong evidence for astrocyte activation induced by cytomix and demonstrate the potential of astrocytes as a neuroinflammation indicator in the µSiM-hBBB.
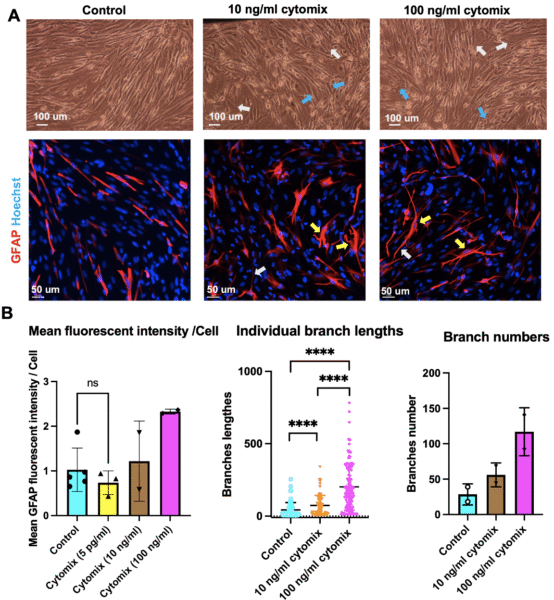
Figure 2 Cytomix-induced astrocyte activation and morphological changes. A) Phase contrast (10 x top) and immunofluorescent images (bottom) of cytomix simulated and non-simulated astrocytes. Simulated astrocytes displayed a more dendritic-like shape, with hypertrophic cells (blue arrows) featuring long branches (white arrows). The GFAP expressed in these cells has also become filamentous (yellow arrows) instead of elongated. B) Immunofluorescence analysis revealed that the expression of GFAP amplified in response to cytomix stimulation, with higher levels observed at higher concentrations. Low doses of cytomix (5 pg/ml) did not significantly influence the GFAP expression compared to non-simulated astrocytes (n = 2 or 3). Statistical analysis of branch lengths (median with interquartile range) and branch numbers (mean with standard deviation) confirmed that both branch length and numbers of astrocytes increased after the simulation (n=2 or 3). *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
Appendix
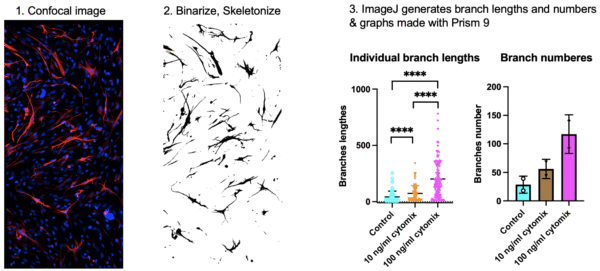
S1 Calculate branches of NHAs using single cell skeletal analysis9 ImageJ
Reference
1 Colombo, E. & Farina, C. Astrocytes: Key Regulators of Neuroinflammation. Trends in Immunology 37, 608-620, doi:https://doi.org/10.1016/j.it.2016.06.006 (2016).
2 McCloskey, M. C. et al. The Modular µSiM: A Mass Produced, Rapidly Assembled, and Reconfigurable Platform for the Study of Barrier Tissue Models In Vitro. Advanced Healthcare Materials 11, 2200804, doi:https://doi.org/10.1002/adhm.202200804 (2022).
3 Hudecz, D. et al. Ultrathin Silicon Membranes for in Situ Optical Analysis of Nanoparticle Translocation across a Human Blood–Brain Barrier Model. ACS Nano 14, 1111-1122, doi:10.1021/acsnano.9b08870 (2020).
4 Hyvärinen, T. et al. Co-stimulation with IL-1β and TNF-α induces an inflammatory reactive astrocyte phenotype with neurosupportive characteristics in a human pluripotent stem cell model system. Scientific Reports 9, 16944, doi:10.1038/s41598-019-53414-9 (2019).
5 Liddelow, S. A. et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature 541, 481-487, doi:10.1038/nature21029 (2017).
6 Pediaditakis, I. et al. A microengineered Brain-Chip to model neuroinflammation in humans. iScience 25, doi:10.1016/j.isci.2022.104813 (2022).
7 Eddleston, M. & Mucke, L. Molecular profile of reactive astrocytes–implications for their role in neurologic disease. Neuroscience 54, 15-36, doi:10.1016/0306-4522(93)90380-x (1993).
8 Pekny, M. & Nilsson, M. Astrocyte activation and reactive gliosis. Glia 50, 427-434, doi:https://doi.org/10.1002/glia.20207 (2005).
9 Green, T. R. F., Murphy, S. M. & Rowe, R. K. Comparisons of quantitative approaches for assessing microglial morphology reveal inconsistencies, ecological fallacy, and a need for standardization. Scientific Reports 12, 18196, doi:10.1038/s41598-022-23091-2 (2022).
Here is the reactive astrocyte paper mentioned in the meeting. https://doi.org/10.1038/s41593-020-00783-4