Using Dual-Scale Membranes as a Model for a ‘Leaky’ BBB
Introduction
Many neurodegenerative diseases, including Alzheimer’s disease and Parkinson’s disease, are associated with an early breakdown of the blood brain barrier (BBB). This includes an increase in permeability and pericyte degradation [1]. We hypothesize that using dual-scale (nanoporous and microporous) membranes in the µSiM-BBB model will create an inherently ‘leaky’ BBB which can be used to model neurodegenerative diseases.
Methods
IMR90-4 iPSC-derived EECM-BMECs at various passages were seeded onto one-slot µSiM devices with both DS and NPN membranes. The devices were cultured for 6 days and then tested for permeability using the sampling permeability assay with Lucifer Yellow dye. Stimulated devices were stimulated with 10 pg/mL of TNF-α, IFN-γ, and IL-1β in the well for about 16 hours before the permeability assay was performed.
Results

While initially it appeared that there was a difference between the permeability of the DS and NPN devices, as the experiment was repeated it became clear that early differences were more likely due to operator error. With the DS devices it is easier to disrupt the EC monolayer by pipetting too quickly in the channel, increasing the permeability. As both operators became more accustomed to using the DS devices we see the permeability converging on a more consistent value which is close to that of the NPN devices, around 0.5 x 103 cm/min, which is the target value for tight barrier. From these experiments we can see that despite the hypothesis, using 3µm DS membranes does not create a BBB which is intrinsically more permeable.
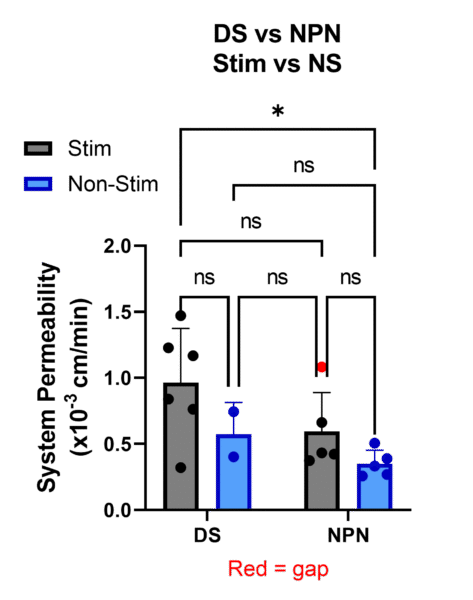
As an alternative method of seeing inherent weakness in the DS devices, the response of the devices to stimulation with inflammatory cytokines (TNF-α, IFN-γ, and IL-1β) was tested. The thought here is that a ‘leaky’ BBB is more susceptible to systemic inflammation such as that seen in sepsis or surgery, so if the DS membrane creates a ‘leaky’ BBB that might be seen by comparing the increase in permeability to NPN membranes. From the data so far, it seems promising that there might be a difference, but from performing a power analysis it was shown that 15 devices/condition will be needed to prove statistical significance. These experiments will be continued to demonstrate conclusively whether there is a difference between the increase in permeability in response to cytokine stimulation of DS devices as compared to NPN devices.

Another way to see an intrinsic difference between the DS and NPN membranes could be to increase the size and/or quantity of the micropores in the DS membranes. A preliminary experiment with 5µm DS membranes did show a difference as compared to NPN membranes, but this will need to be repeated to show that there is a difference between the two conditions. If the 5µm devices turn out to not show a difference, increasing the quantity of micropores could also be investigated.
Conclusions
Using the 3µm DS membranes in µSiM devices does not create an intrinsically more permeably barrier, but they may create a barrier which is intrinsically weaker in response to cytokine stimulation which could still be used as a model of a ‘leaky’ BBB for our studies. Alternatively we can look into increasing the size and/or quantity of micropores until reaching a point where the BBB is more permeable even without the cytokine stimulation. While there are promising preliminary results with the 5µm DS membranes, they need to be repeated to confirm that they reproducibly create a ‘leaky’ BBB.
References
- Sweeney, M.D., A.P. Sagare, and B.V. Zlokovic, Blood-brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat Rev Neurol, 2018. 14(3): p. 133-150.