Gap Junction Formation Across SiO2 Nanomembranes
Background
The purpose of this experiment is to investigate the formation of gap junctions between co-cultured HUVEC and ADSC across SiO2 membranes. Gap junctions are specialized intercellular connections through which nutrients and small molecules can be transported. HUVEC will be stained with CFDA (green) and ADSC will be stained with NG2 (red) . Gap junction formation will be identified by ADSC that are stained both green and red.
Methods
Seeding HUVEC
- CytoVu devices were assembled in 24-well plates using PDMS gaskets cut using the craft cutter and SiO2 membrane gaskets. In this experiment, 0.5µm-pore high porosity SiO2 membranes were used.
- After assembling the devices, the plates were cured overnight at 73C. Before cell culture, the plates were exposed to UV light, ~15 min. each side.
- 1% Geltrex was prepared and 20µL were added into each of the large bottom PDMS gaskets. The plates were then closed and flipped and left at RT inside the hood for ~30 mins.
- The Geltrex was removed using a micropipette and then 20µL of 1% Geltrex was added on top of each membrane. The plates were covered and left at RT inside the hood for ~30 mins.
- After the 30 mins., the Geltrex was removed.The plate was let to dry inside the hood with the lid partially on.
- HUVEC were then seeded under the SiO2 membranes inside the large PDMS gasket at a seeding density of 17,500 cells/cm2 . A tissue was wet using distilled water and placed on the inside of the lid. The plate was then closed with the tissue facing the inside of the plate and the plate was flipped and incubated for ~45 min. at 37C.
-
After that time, the plate was returned to normal position and the wells were flooded with warm HUVEC media. Plates were incubated at 37C for 3 days until HUVEC reached confluency.
Staining HUVEC
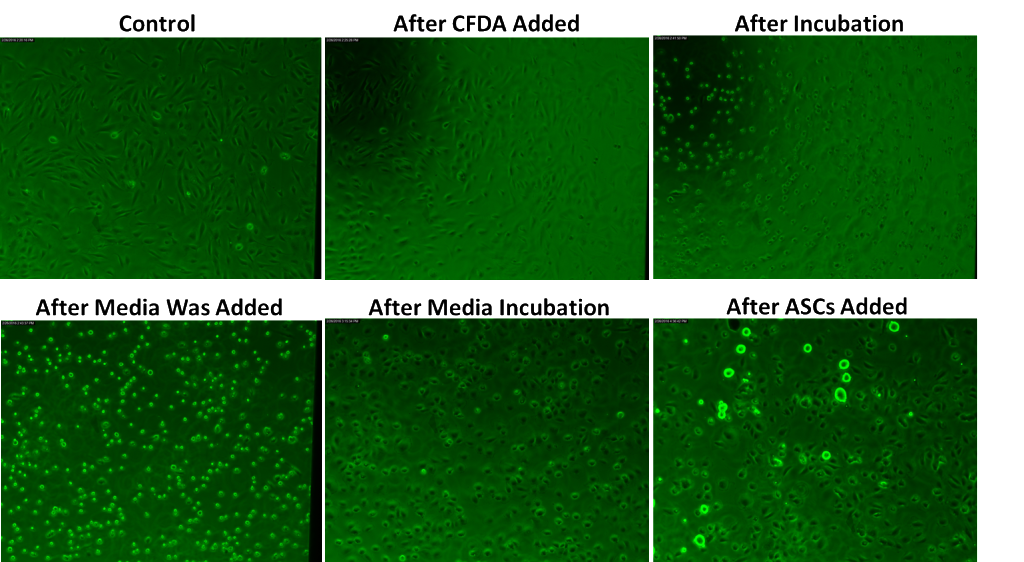
- After three days of incubation, the HUVEC were stained using CFDA working solution and then incubated at 37C for ~15 min.
- The cells were washed with PBS and then flooded with HUVEC media and incubated at 37C for ~30 min.
Seeding ADSC
- After that time, ADSC were seeding at a seeding density of 17,500 cells/cm2. The cells this time were added on top of the SiO2 membranes.
- The plates were covered and incubated for ~10 min. at 37C.
-
After that time, the wells were flooded with HUVEC media. The plates were incubated for 3 days until ADSCs reached confluency.
Staining ADSC
- The ADSC were stained using NG2 and were incubates at 37C for ~30 min.
- The wells were then washed with PBS 2x and were flooded with HUVEC media.
Imaging
- Images were obtained using phase contrast, GFP, and TxRed fluorescent channels.
- Each SiO2 gasket could be imaged entirely with a total of 4 10x images, one of each corner.
- Images were overlaid and analyzed to look for gap junctions.
Data
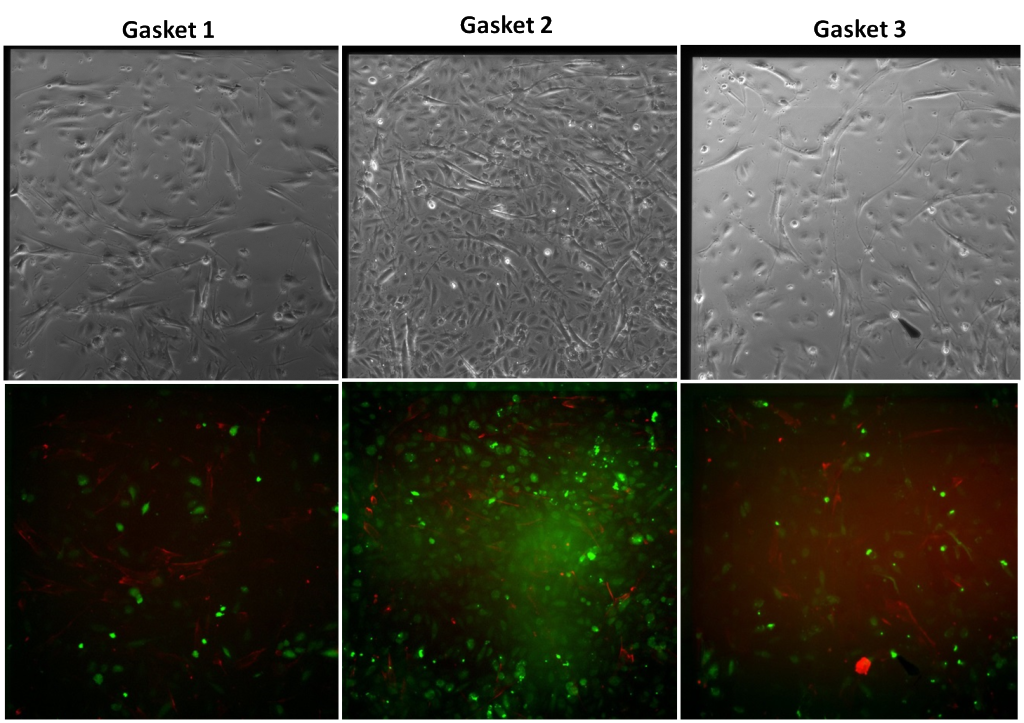
Trial 1
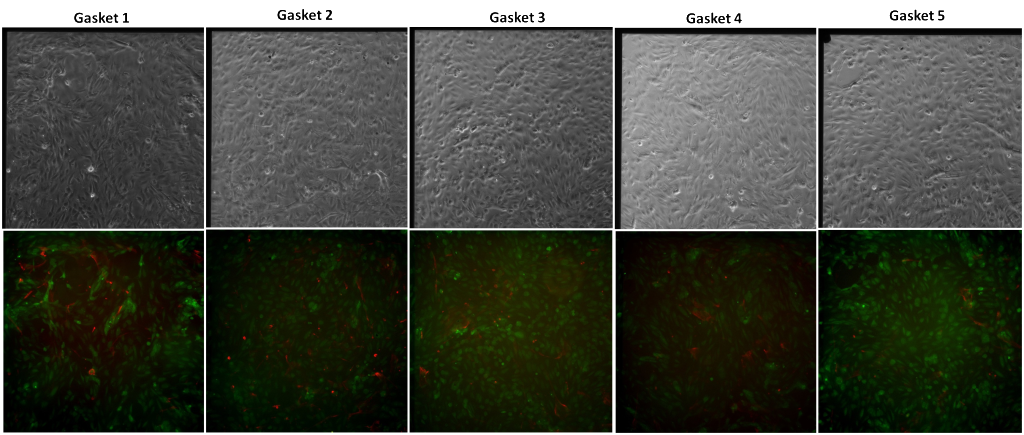
Trial 2
Observations
In Trial 2, gap junctions between the HUVEC and ADSC cultures across the SiO2 membrane were not found. One of the six gaskets broke but the cells did not fall off the SiO2 gaskets given that it was found that the HUVEC were disrupted after the CFDA incubation. To keep them from detaching from the membrane, the CFDA stain was not washed off by pipetting PBS under the membranes. Instead, the wells were washed through PBS flooding, allowing the cells to reattach to the surface.
Conclusion
Gap junctions were not found in any of the trials. One possible reason could be that the staining protocol is not effectively staining gap junction formation but this technique has been used previously in other studies. Another possibility is that the 1% Geltrex coating on both sides of the membrane is blocking gap junction formation.