ASAIO notes
ASAIO /əˈsī ō/ noun
- American Society of Artificial Internal Organs. There is an associated Journal, “ASAIO Journal”, and a yearly conference for the presentation and discussion of research relating to advances in medical device technology. While the initial focus of the society was on renal devices, the artificial kidney, currently the majority of work by ASAIO members is cardiovascular including the popular Left Ventricular Assist Device (LVAD).
At this year’s ASAIO conference, held on June 15 – June 19 in San Francisco, there were three devices or systems along with supporting areas of research presented and some interesting work on vascular access. We should be familiar with most of these by now.
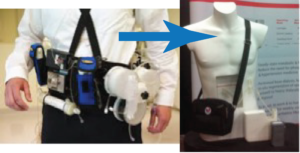
- Automated Wearable Artificial Kidney(AWAK), UCLA, Dr. Victor Gura, presented by Martin Roberts, PhD, AWAK Technologies Inc, North Hills, CA
This is a miniaturized Peritoneal Dialysis (PD) device. The latest device format is impressively small and well contained. There is a disposable filter/dialysate cartridge. Not FDA approved yet.
Operation is as simple as three button presses, ‘On’, ‘Prime’, and ‘Start’. This is great for patients using PD. PD uses a patient’s peritoneum as the filter with dialysate cyclically pumped into and out of the abdomen. According to some nephrologists I’ve spoken with, the average time on PD before infection (peritonitis) renders it useless and necessitates a switch to Hemodialysis (HD) is 18 months. This is great for those awaiting a kidney transplant of as a bridge to the longer lasting HD. (Remember the life expectancy for dialysis patients is 3.5 years.)
- Bioartificial Kidney (BAK), University of Michigan, David, MD
In this device, the first filter performs plasmapheresis (like the glomerulus), plasma is separated from the whole blood, both are passed to the renal assist device (RAD), a filter with kidney cells grown on the luminal side of the hollow fiber membranes. The second filter allows for active transport of needed molecules (e.g. glucose, and salts) back into the blood (like the proximal tubules).
Cells used in polymer membranes. Started with flat membrane will need to move to hollow fiber…
Proxima tubule Epithelial cells Collagen Poly l-Dopa PES membrane
They tested with inulin (5 kDa) and creatinine (113 Da). Inulin should not go through with cells, creatinine should just based on size. This didn’t really show anything that the images of tight junctions showed, that cells grew to confluence. Another group (Netherlands) showed that they were able to turn off active transport but this test only showed sized-based filtration with cells.
The cells have been shown to last up to 2 weeks on hollow fiber but they have done no other (successful) experiments.
They believe no collagen will be needed with the right topography (polystyrene)
Water balance, in the kidney done by the loop of Henle and the Sodium gradient in the surrounding kidney tissue, but is not specifically addressed by BAK. The epithelial cells in the kidney differentiate and contain different active transporters depending on there proximity to the glomerulus.
This is one of many groups growing, or attempting to grow epithelial cells on hollow fiber membrane filters. There goal is a wearable bioartificial kidney (WeBAK)
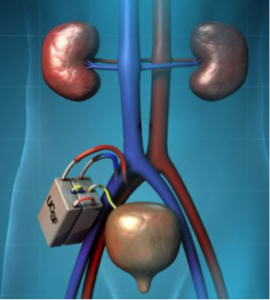
- Implantable Artificial Kidney (aka Implantable Renal Assist Device (IRAD)), Vanderbilt, Shuvo Roy, PhD, William Fissell MD, presented by Dr. Fissell and Benjamin Feinberg, PhD, UCSF.
Dr. Fissell /fi ˈzel/ delivered a rhetoric filled speech about the ‘moonshot’ of the implantable artificial kidney. Attempts at improving extracorporeal filtration will always fall short of the target of providing complete kidney replacement. Demand for transplantable kidneys (from either living or cadaverous donors) is rising yet the supply remains flat. This is true. He did not present the recent work showing thrombosis, nor did he address how water balance would be handled. I suppose they think that if they have proximal tubule endothelial cells, they will have the active transporters needed, but they won’t. PTECs contain different transporter populations based on their proximity to the glomerulus (as I leaned in the presentation regarding bioengineered proximal tubules.
(NOTE: They are also working on BIOARTIFICIAL PANCREATIC ISLET SYSTEM (BPICS)
A treatment of insulin-dependent diabetes, the BPICS will use pancreatic islet cells to regulate blood sugar.)
- Others…
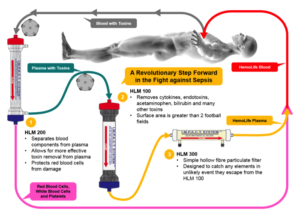
a. Coupled Plasma Filtration and Adsorption: The IMPACT System. Patrick Maguire, MD, PhD, MBA, Hemolife Medical, Dana Point, CA
b. A Portable Hemofiltration System Newly Designed with a Small Centrifugal Pump
- The main import of this is the use of the centrifugal pump in place of a peristaltic roller pump.
- They claim low hemolysis
- Carbon Block Column for Regeneration of Dialysate in CVVHD Stephen Ash, MD, HemoCleanse Technologies LLC, Lafayette, IN
Continuous Venovenous Hemodialysis (CVVHD)
-Carbon, embedded in a polymer matrix that allows toxins to access the majority of the carbon’s surface area was presented. Plasma is passed through the column and toxins as absorbed by the carbon
- Mixed Matrix Membrane Dimitrios Stamatialis, PhD, University of Twente, Enschede, Netherlands
Mixed matrix membranes for removal of protein bound toxins.
http://www.ncbi.nlm.nih.gov/pubmed/23876759
- Bioengineering of a Proximal tubule
(Aside: Anglerfish don’t have glomerulus (plasma filtration), only tubules so… “Is the glomerulus really needed?” )
Conditionally Immortalized Proximal Tubular Epithelial Cell (ciPTEC)
The cells loose transporters when they are grown in hollow fibers so they regulate them back in somehow. Not sure.
This is the Dutch group that have tested PTECs for passive and active transport of molecules and have successfully turned off the transport showing that active transport was actually being done by the cells.
One hypothesis of this group: Protein bound toxins such as phenols and inoles will be removed actively by the epithelial cells.
NOTES:
AWAK is a peritoneal dialysis and not a competition to our work unless they can overcome the infection problems with catheter ports.
All ‘artificial kidneys’ implantable, wearable, or standard need to grow epithelial cells (at least) to replicate renal tubule functions. This perhaps is mature enough for extracorporeal filtration but not even close for implantable. Implantable is also problematic due to ultrafiltration, and regulation of filtration rate, which is affected by MAP and MVP. All of these problems have solutions, they just aren’t there yet. They also want to include remote data so they will need a battery, recharging system, and associated electronics. Luckily the human abdomen has plenty of room.
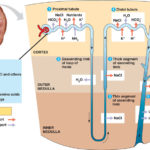
I’m including a good cartoon of a nephron to show its many functions, some of which are being targeted by the above devices.