Preliminary potentiostat measurements on iBidi format devices
Thanks to Sidahmed Abayzeed and Emilia Moradi.
Summary:
I’ve started making impedance spectroscopy measurements with a potentiostat in the iBidi devices. The data is noisy, but there is a clear change in the curve characteristic as cells grow upon the nanomembrane. NPN seems to be working better than MgF2, for these measurements.
I’ve measured the impedance of nanoporous membranes (MgF2 and NPN) in two different geometries, both within the iBidi format and the Ussing Chamber, with 5M NaCl and 1X PBS. Using 2 gold reference electrodes, we can inject current and measure the voltage across the nanomembrane, but the potentiostat goes further (tries to keep the voltage the same value) and can separate the resistive and capacitative components from the system, allowing us to generate measurements of the frequency behavior of the whole system (magnitude and phase). The technique is known as Electrochemical Impedance Spectroscopy (EIS). The goal of these characterizations is to establish a baseline measurement for use in future experiments with cells, analyzing the amount of impedance the different elements contribute to the overall behavior.
iBidi Device EIS Measurements:
The basic principle is to measure the frequency dependent impedance of the entire system, then model that system using an equivalent circuit.

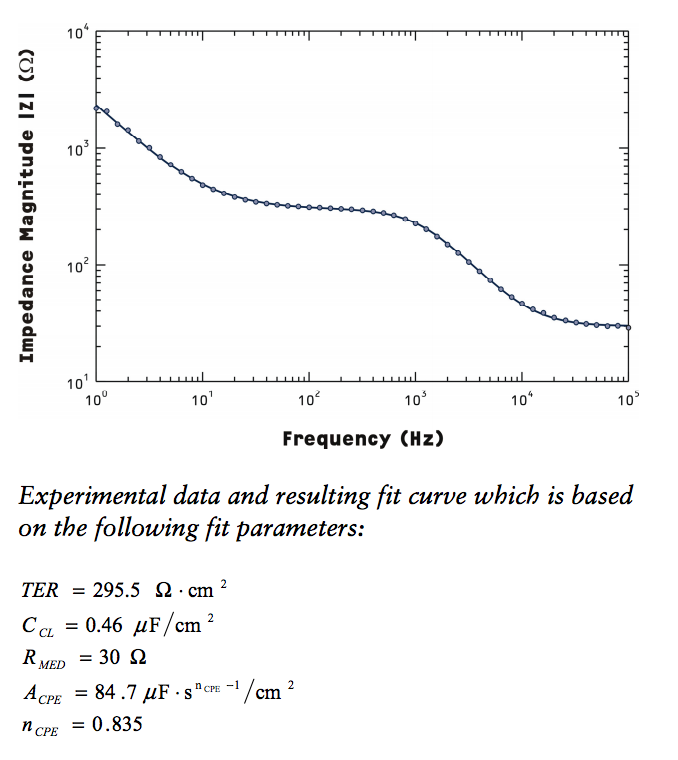
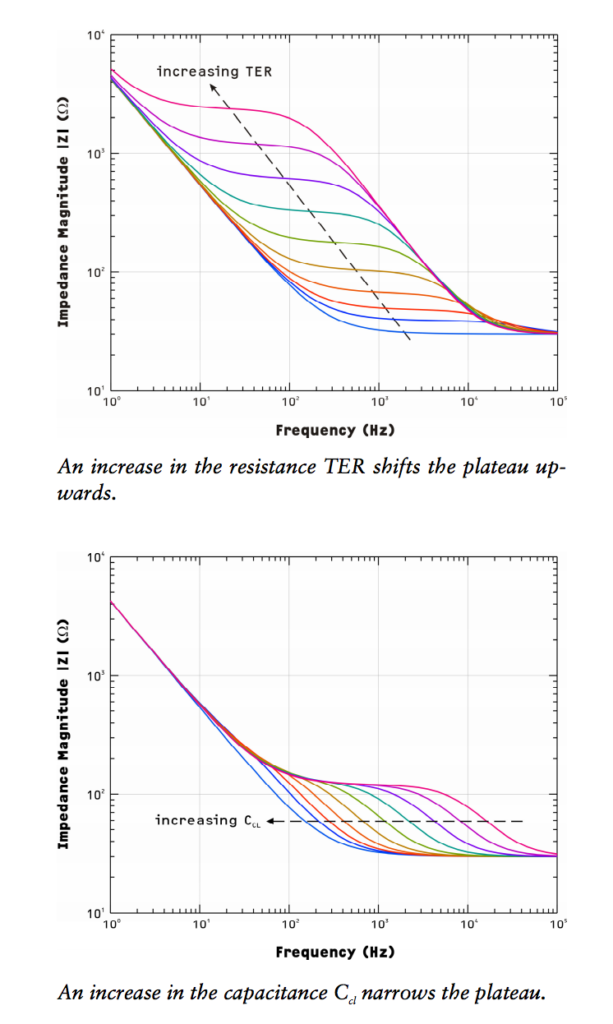

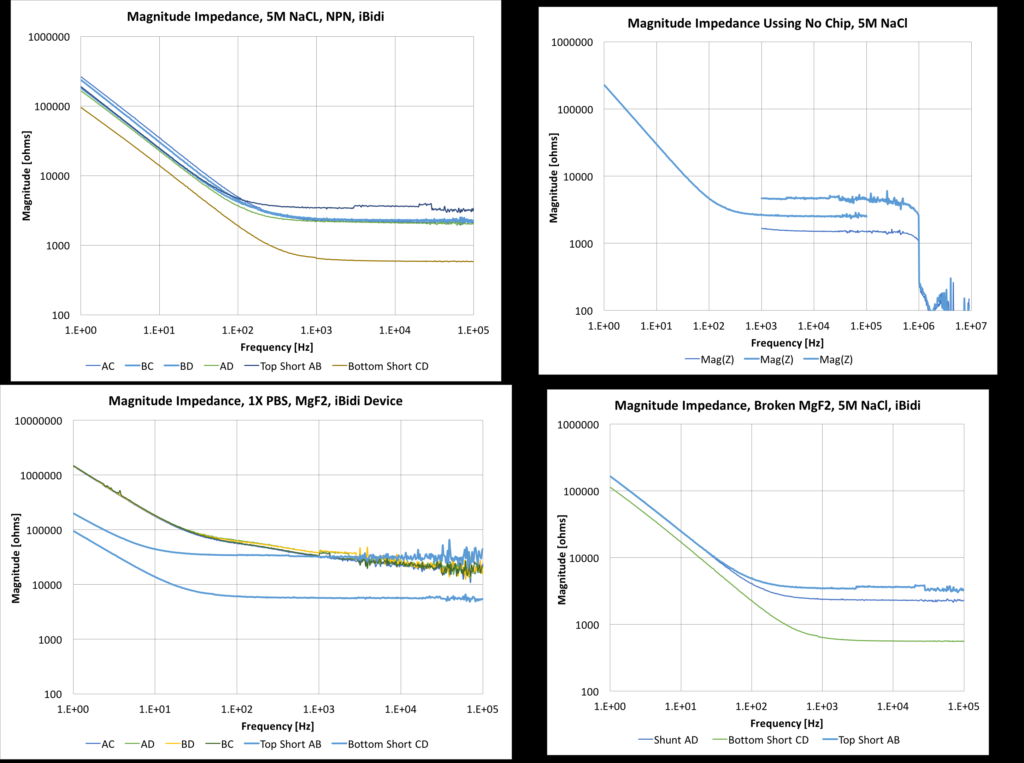
This montage shows a number of devices at different salt strengths. Even with the highest salt available, the iBidi device is about an order of magnitude more resistive than the commercial z-scope. This will only worsen as we use cell media, which is much less conductive. The lowest resistance path is the bottom short and the highest resistance path is the top short (due to channel geometry in the ibidi device). The MgF2 substrate has an odd behavior, showing clearly two slopes (meaning two time-constants -> two capacitances), which I need to validate with further experiments, as well as being more resistive than the top short. Speculating a bit, this could mean that the pores are blocked, or narrow/few enough to actually provide electrical resistance. It could be also a feature of the geometry of the pores themselves, being a different shape than the pnc-si.
There are a few more configurations still to run, primarily with a non porous membrane, as well as MgF2 at high salts. So far, the baseline impedance is two orders of magnitude higher than a commercial system, but the impedance level appears to be stable from device to device.
Cell seeded Membranes:
Cells were seeded on Fabricated iBidi devices
- Spread Norland-61 optical adhesive on iBidi device to adhere chip, bottom and top coverglasses.
- Membranes were mounted with the flat side facing down (minimizes working distance), using the same adhesive.
- Cure with UV light (365nm) for 2.5 hrs (bottom side up first, 1.5 hours)
- Sterilize with Ethanol, 1 hr
- Filled all the channels and then submerged entire device.
- Dry devices, replace Ethanol in channels with Media, 10 min
- It’s easy to tell when the membrane is broken, because the fluid no longer stays in the appropriate channel; filling the top channel causes the fluid to rise in the bottom channel ports.
- Coat with PLL, 10 minutes as an adhesion promoter. Rinse 3x with media.
- Seeded 100,000 Calu-3 (lung epithelial) cells in the bottom channel, capped all channel ports, then inverted and placed in incubator.
- I’m using Calu-3 instead of ARPE-19 because the cells form a really strong barrier (10-50x better than RPE).
- Inspection revealed about 20-30 cells below the active area, before inversion.
- Upon inspection, it seemed that the Calu3 cells were having difficulty adhering to NPN, even with the PLL coating.
The cells were grown for 72 hours in the incubator (35 C, 5% CO2, 90% RH). In a parallel effort, cells were seeded on coverglass with the fully cured Norland 61 resin, to see if there were obvious deleterious effects. The device was measured before the cell media was exchanged, and then 1 hr after the cell media was exchanged, to allow the cells to equilibrate.
While the initial device failed after washing, another device was cultured up to 16 days using the same protocol.
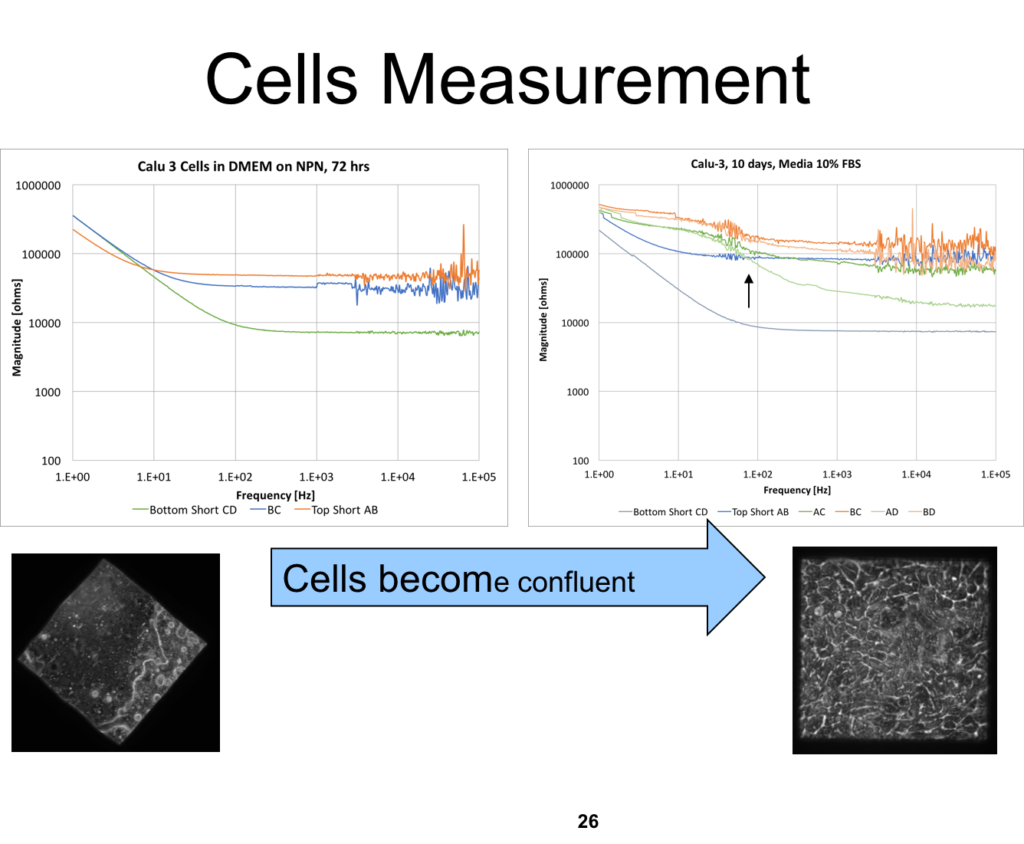
The measurements are very noisy at the higher frequencies, and we aren’t sure what is causing this behavior. The noise also appears to be exacerbated at higher baseline resistance values. There seems to be another band of noisy measurements around 50-100 Hz too.
Ideally we’d like the impedance of the cell layers to roughly match the impedance of the device, which would increase the sensitivity of the measurements, as well as move the electrodes closer to the substrate, minimizing background resistance. Kevin’s design from a few years ago is a step in the right direction.
There’s a lot of work ahead in the next month, but these results are encouraging.