Issues in achieving Neutrophil migration through collagen gels
For many months, Emma and I are trying to achieve neutrophil migration through endothelial cells and through the underlying collagen gel. I need to explain the details of my device first. The schematic is shown as below.
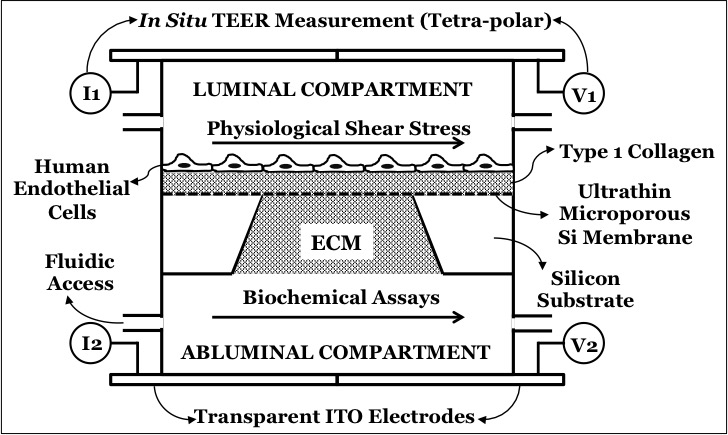
I am using a 3 um porous membrane to allow neutrophils to cross the HUVECs in the collagen gel below the membrane. The top gel, however, is for a different reason. In the past, I had difficulties in retaining the cells on the surface under the influence of shear stress. The thickness of top gel is 100 micron, while the bottom gel is about 600-micron gaskets. However, the top gel seems to get compressed as soon as any normal or tangential force is applied to the gel, due to its compliance. Look at the picture below of the HUVECs before and after exposed to flow.
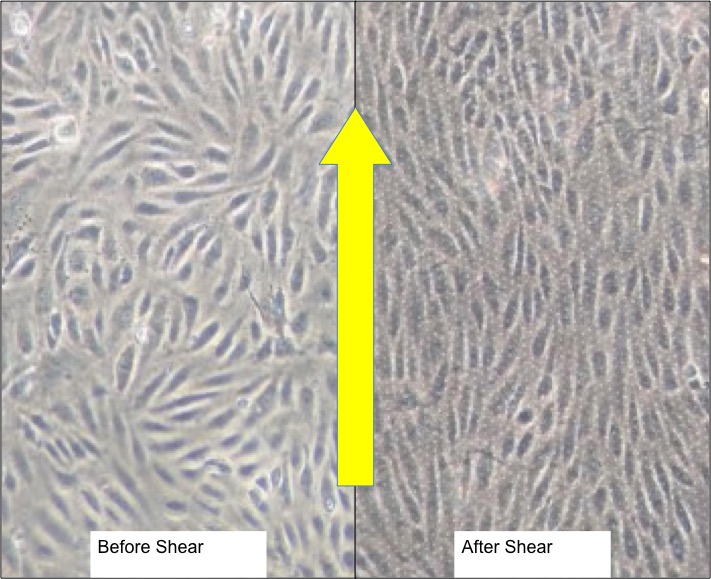
This compression of the collagen gel essentially increases the height of the flow channel, thus significantly increasing the flow rates’ requirements to maintain the same shear stress of 10 dynes per sq.cm. Increasing the gel concentration can increase the gel stiffness, but we are already at the upper end of the physiological range of concentration. The pore size will also reduce further if the concentration gets any higher.
The compressed top gel is also an issue for neutrophil migration, the reason for this post! Check the following sem images.
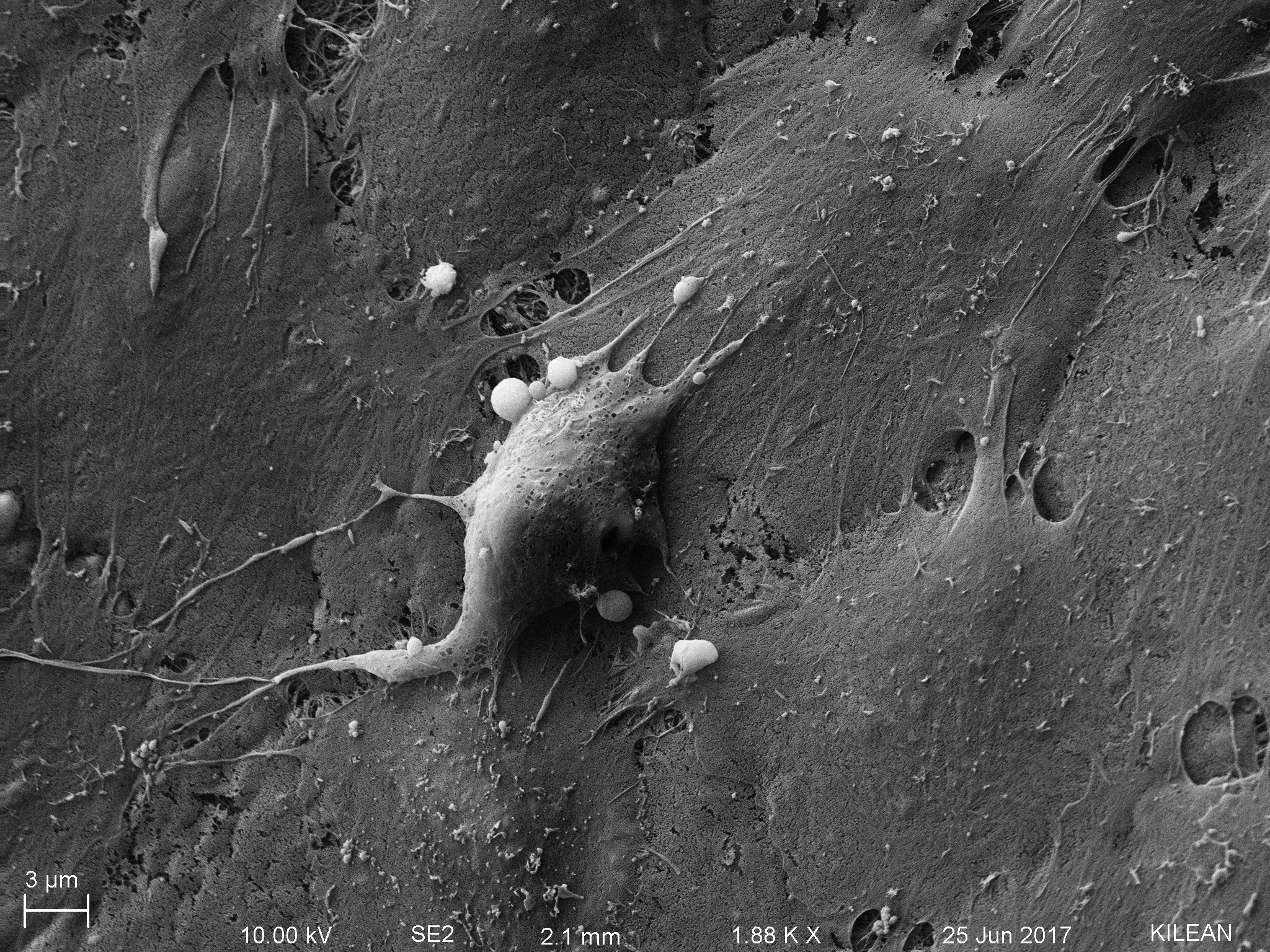


PC: Kilean Scott Lucas!!!
Looking at the scale bars, one can understand the relative size of neutrophils compared to the mesh size of the collagen gel. Rick Waugh cautioned that just because the mesh looks dense doesn’t imply that it is impermissible for neutrophil migration, but still the size difference between gel-mesh and neutrophil body is stunning.
Please note that neutrophils are activated and are motivated to transmigrate, as observed in the following picture.

We later realized the issue was not just the gel compression, but the type of gel itself. Last winter, when I first started working with collagen gel, I did achieve successful transmigration. I had bottom gel only, and no endothelial cells on top and neutrophils were observed in the bottom compartments. So I deiced to repeat the exact same setup from the last year to reproduce the results. I was using collagen from a different company back then called as Enzo life sciences. After I ran out of the gel, I switched to Corning collagen. The two proteins are derived from same rat tail, but their gelling protocols (stoichiometry) are slightly different. Anyways I went back to old collagen, which was still left in tiny amounts, and repeated the same experiment. Since I was working with neutrophils only back then, both neutrophils and fMLP were suspended in HBSS+Ca+Mg, and there was an only bottom gel. The latest work involved HUVECs; so all the cells and reagents were in EC medium MCDB-131, and there were both bottom AND top gels. The summary of my experiments is shown below.
Summary of experiments with Corning gel
| Media | Bottom Gel | Top Gel | Results |
| MCDB | Yes | Yes | 🙁 |
| MCDB | No | Yes | 🙁 |
| MCDB | Yes | No | 🙁 |
| HBSS | Yes | No | 🙁 |
Summary of experiments with Enzo LS gel
| Media | Bottom Gel | Top Gel | Results |
| HBSS | Yes | No | 🙂 |
| MCDB | Yes | No | 🙂 |
| MCDB | Yes | Yes | 🙁 |
Essentially, I couldn’t achieve any migration with Corning collagen using MDCB media or using HBSS media (which worked for me in the past).
With Enzo LS collagen, results are mixed. The media seems to be a neutral factor. I observed successful migration with both MCDB and HBSS media when there was ONLY bottom gel present. However, when both top and bottom gels are present, this observation was defied. This is very critical, because, in the absence of the top gel, it is impossible to keep the cells attached on the surface of the membrane. Jim hypothesized that the ‘collapse’ of the top gel makes it denser for neutrophil entry and migration in it. Once the cells are in the top gel, they also have to look for an open pore to go to the bottom side to follow the gradient. The schematic looks like below.
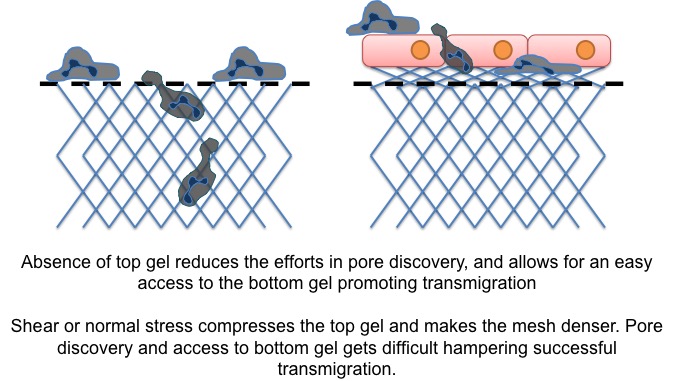
Or maybe, the gel need not be dense enough to block the neutrophils from migration, but once they are inside the gel, pore discovery might be an issue. Either the reason, the inability to achieve interstitial migration is very detrimental to my project. That brings to the big question: what EXACTLY I need to achieve?!?
My hypothesis is to test whether the breach of the basement membrane by and during neutrophil extravasation affects the permeability or not. So TECHNICALLY, I don’t care what happens in the interstitial collagen space, as long as the basement membrane (BM) migration is still going on. However, testing whether the basement membrane is breached or not (by integrin blocking) is easy to decide, by observing interstitial migration. Neutrophils HAVE to breach BM to enter collagen gel. So even if the neutrophils breach the BM secreted by HUVECs cultured on the top gel, it is difficult to say conclusively whether or not the BM is actually breached or not. If HUVECs are grown directly on a flat membrane, then the presence or absence of neutrophils in the bottom collagen gel is a definitive proof of whether or not the BM is breached or is retained intact. Having said that, the absence of top gel makes attachment-under-shear an issue and can compromise the utility of our proposed device.
Summarily, the potential outcomes and hypothesis-testing can be summarized as follows.
| Configuration | Observation | Conclusion |
| WITH Top Gel | Integrin-blocked neutrophils are not seen in the bottom gel | BM is intact, or maybe the BM is breached but neutrophils are not able to enter the top gel below BM, and hence absent in the top gel |
| WITHOUT Top Gel | Integrin-blocked neutrophils are not seen in the bottom gel | BM is intact, ONLY IF the control untreated neutrophils are seen in the bottom gel |
I will be running a comprehensive experiment next week with HUVECs grown on chip directly with bottom gel, and checking for neutrophil migration through HUVECs and through the collagen gel. Stay tuned!