Extended Endothelial cell Culture Method-differentiated Brain Microvascular Endothelial Cell-like cell (EECM-BMEC-like cell) growth on Dual-Scale Membranes
Introduction
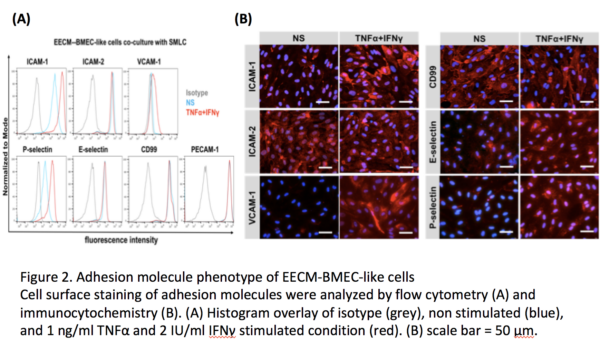
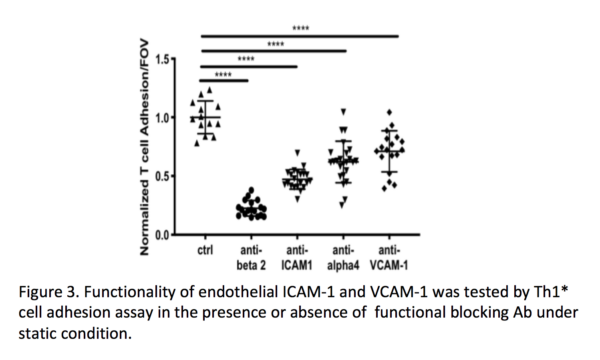
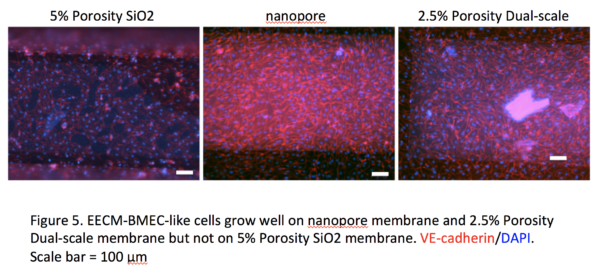
Human induced pluripotent stem cell (hiPSC)-derived blood-brain barrier (BBB) models have proven to be useful to study disease pathomechanisms and drug delivery into the central nervous system. Currently available hiPSC-derived BBB models are well characterized with respect to diffusion barrier properties and specific transporters, however not optimal for studying BBB-immune cells interactions because of lack of key adhesion molecules. In our lab, we recently established a novel protocol to differentiate hiPSCs to BMEC-like cells, namely the Extended Endothelial cell Culture Method (EECM) with good barrier properties and mature tight junctions (Figure 1). Importantly, EECM-BMEC-like cells exhibited constitutive cell surface expression of ICAM-1, ICAM-2 and E-selectin. Pro-inflammatory cytokine stimulation increased cell surface expression of ICAM-1 and induced cell surface expression of P-selectin and VCAM-1 (Figure 2). Coculture of EECM-BMEC-like cells with hiPSC-derived smooth muscle-like cells or their conditioned medium further increased induction of VCAM-1. Functional expression of endothelial ICAM-1 and VCAM-1 was confirmed by T-cell interaction with EECM-BMEC-like cells (Figure 3). We further verified their capability to support immune cell migration by investigating T-cell interaction with EECM-BMEC-like cell monolayers under physiological flow using commercially available Ibidi dish (Video 1). In this in vitro flow assay, we could analyze detail immune cell post arrest behavior, however cannot collect immune cells, which have migrated through EECM-BMEC-like cell monolayer because of the lack of two compartments with large pore membrane. Furthermore, we failed to cultivate EECM-BMEC-like cells onto commercially available polycarbonate 3μm pore Transwell filter (Costar 3415) (Figure 4). Cultivating EECM-BMEC-like cells onto Dual-scale membrane allow us to study detail phenotype of migrated immune cells. This post summarize first trial if EECM-BMEC-like cells grow well onto Dual-scale membrane.


Method
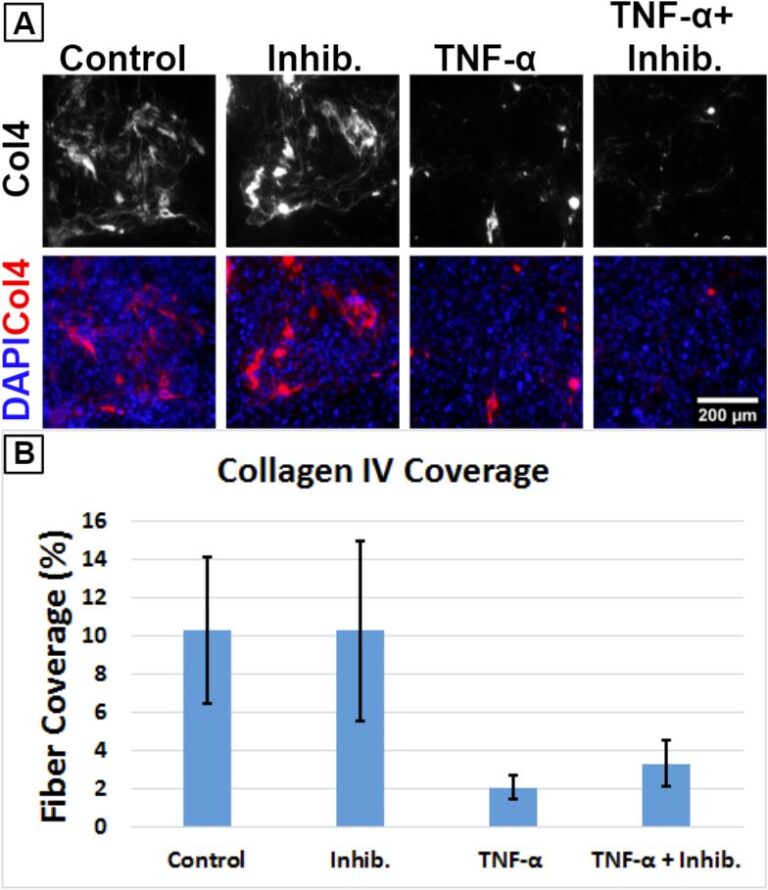
Nanopore membrane, 2.5% Porosity Dual-scale membrane, or 5% Porosity SiO2 membrane was assembled using Trench-Down device and let the device sit under UV light for 20 minutes. Membranes were coated with 400 ug/ml collagen Ⅳand 100 ug/ml fibronectin for both side for 1hr at 37°C. After 1hour, excess collagen/fibronectin were removed and membrane was washed with hECSR medium (human endothelial SFM (ThermoFisher) supplemented with 1 × B27 and 20 ng/ml FGF). hECSR medium was added to bottom channel and 40,000 cells/cm2 (15,000 cells/100 μl) EECM-BMEC-like cells from healthy control 1 clone 2 (HC1_2) were seeded and incubated at 37°C, 5% CO2 for 2 hours. After 2 hours, hECSR medium was changed in the top well and bottom channel. hECSR medium was changed every day for 4 days. After 4 days of culture, EECM-BMEC-like cell monolayer was fixed with 1% PFA (10 minutes, RT). After washing, cells were blocked and permeabilized with 5% skim milk containing TritonX. Cells were incubated with mouse anti-human VE-cadherin Ab (SIGMA, clone F-8) for 1hour at RT. After washing, cells were incubated with Cy3 conjugated goat anti-mouse IgG for 1hour at RT. After washing, fluorescein microscopy images were taken.
Results
Conclusions
We have successfully grown EECM-BMEC-like cells onto Dual-scale membrane using one healthy control clone. Pore density seems most important factor for growth of EECM-BMEC-like cells. Next step would be check if we can grow EECM-BMEC-like cells from different donors/clones since growth rate is different depends on clones. We further need to evaluate if EECM-BMEC-like cell keeps same phenotype when they are cultivated onto Dual-scale membrane (e.g. tight junction proteins and adhesion molecule expression and permeability to small molecule tracers).