HUVEC polarity to TGF-β1 treatment
Introduction:
Transforming Growth Factor -β1 (TGF-β1) is a key player in the induction and progression of fibrosis in many tissues. This factor has also been established as a regulator in the development of fibrotic diseases for its involvement of different signaling pathways and cross-talks between many signaling cascades. We are in collaboration with Dr. Hani Awad from the Center for musculoskeletal Research (CMSR) to develop an in vitro model to study fibrosis and inflammation in tendon injuries. Most of their work has been done in mouse models of tendon fibrosis where they have been investigating ways to diminish the accumulation of scar formation due to fibrosis in tendons post-injury (Fig.1).

Methods:
Our proposed Tissue-on-Chip for tendon fibrosis and inflammation is composed of a basal vascular layer and a fibroblast-laden hydrogel layer on the apical side (Fig.2). For the ToC experimental set-up, we introduce 10ng/mL of TGF-β1 tenocyte media into the top chamber of the device and non-treated HUVEC media into the bottom channel of the device. The characteristic and morphological changes are described in Fig.2 c) where the fibroblast undergo differentiation into their activated form (myofibroblast) where they secrete extracellular matrix components and recruit immune regulators. The HUVEC monolayer has been shown to undergo various morphological changes upon exposure to TGF-β1 including differentiation into migratory phenotypes (observed during endothelial to mesenchymal transitions) and decreased permeability characteristic of inflammatory phenotypes.

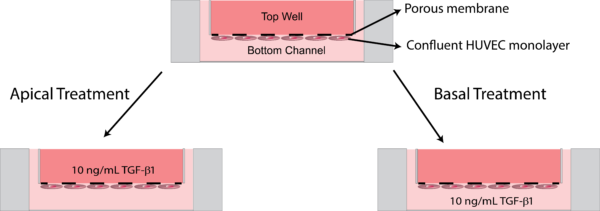
For this experiment, we wanted to study the effects of TGF-β1 treatment on the HUVEC monolayer in the absence of the fibroblast-laden hydrogel in the apical chamber. We grew a HUVEC monolayer at a concentration of 50,000 cells/cm2 in the bottom channel for three days to form a confluent monolayer. The monolayers were then treated on day 4 with 10 ng/mL of TGF-β1 introduced either directly to the top well or to the bottom channel (Fig. 3). after 48 hours of treatment, the layers were fixed with 4% paraformaldehyde and stained with Actin-phalloidin and α-SMA. As an additional control, we performed the same experiment on a HUVEC monolayer grown on glass slides.
Results:
Glass Slide results:
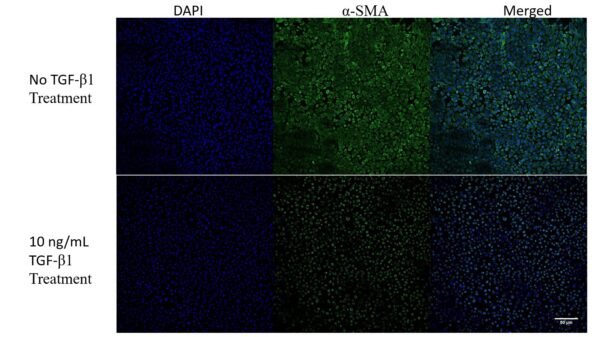
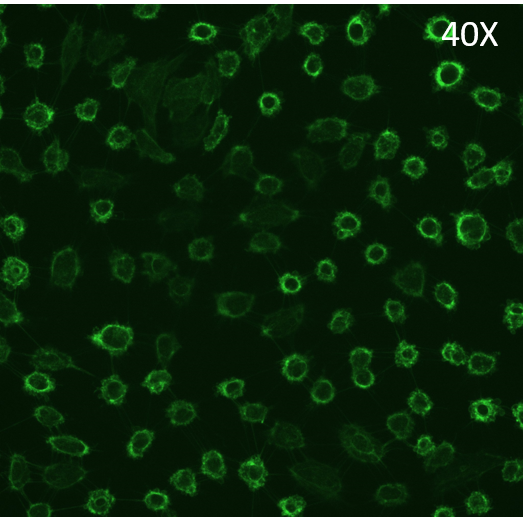
There is certainly an effect on the HUVEC monolayer upon treatment with TGF-β1. There are definitive morphological changes in the cells, yet there does not seem to be a large loss in cell density.
NPN membrane results apical treatment:
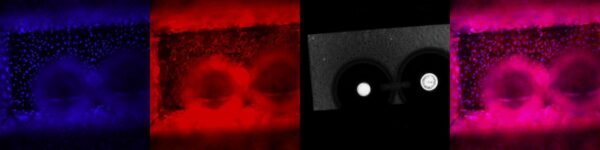
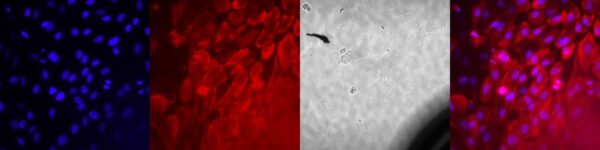
NPN Membrane Results Basal Treatment:
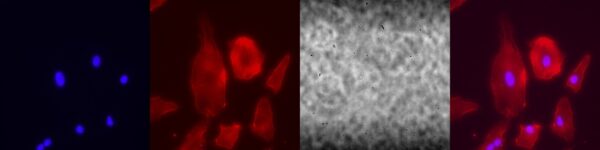
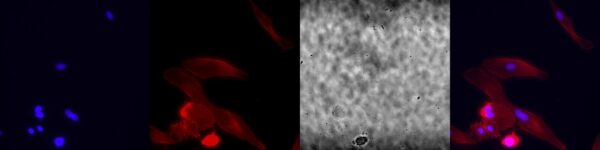
0.5 micron slotted membrane:

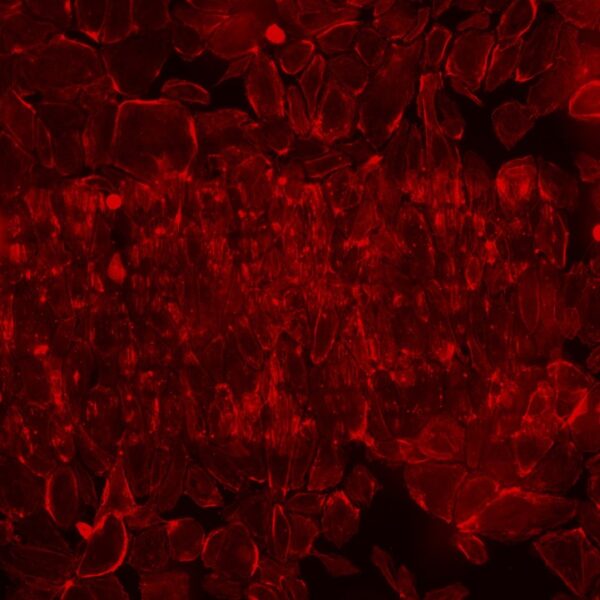
The HUVECs seem to align along the slits. There are large effects upon treatment.
Previous experiments where treatment was introduced apically show drastic morphological changes to the HUVEC layer.