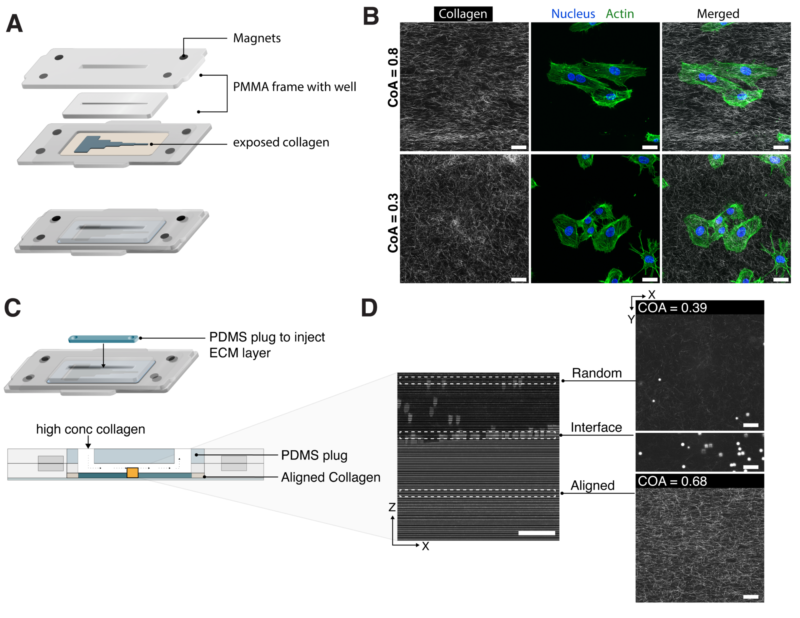
Microfluidic control over millimeter scale alignment of collagen fibers
tissue. At the cell scale, alignment guides cell behavior such as motility, gene expression, and morphology. At the millimeter scale, fiber alignment regulates tissue morphogenesis, facilitates cell-cell communication via force transmission through aligned fibers, and results in anisotropic mechanical properties in tissues. The degree of fiber alignment is also an essential property of type I collagen matrices. Changes in the degree of alignment have been shown to regulate focal adhesion formation, cell motility, and polarization. Given the length scale and wide range of alignment observed in vivo, microfluidic platforms have been used to establish control over fiber alignment in vitro. Early studies to control fiber alignment within microfluidic channels have relied upon shear that is induced during laminar flow. Lee et al. first demonstrated the alignment of a self-assembling collagen solution flowing through microchannels up to 100μm in width and 40μmin height. They showed that collagen fiber alignment was a function of channel width, and no alignment was observed for channels wider than 100μm. While shear has been a common method to induce alignment, the resulting hydrogel constructs are <100μm in width, and <50μm in thickness. Other studies have used microscale flow physics to fabricate free-standing, 3D collagen structures with fiber alignment such as sheets, or to extrude fibers as in bioprinting. However, the resulting structures are free-standing and are not integrated within a microfluidic system, and thus do not offer the versatility and control that is afforded by microfluidics over the microenvironment.
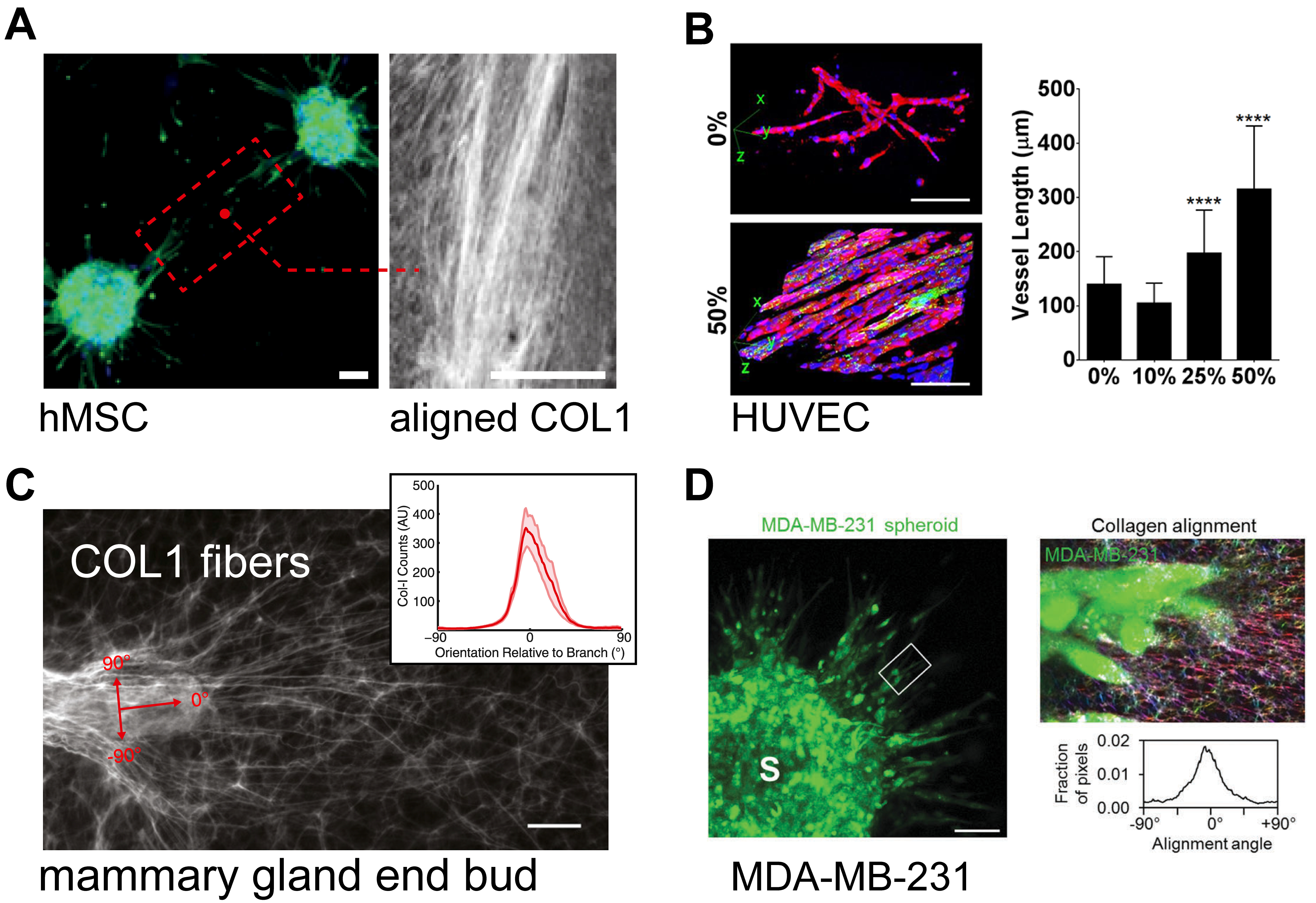
Shear flow did not induce 3D, long-range fiber alignment

Fabrication of segmented channel to induce extensional strain in a self-assembling collagen solution
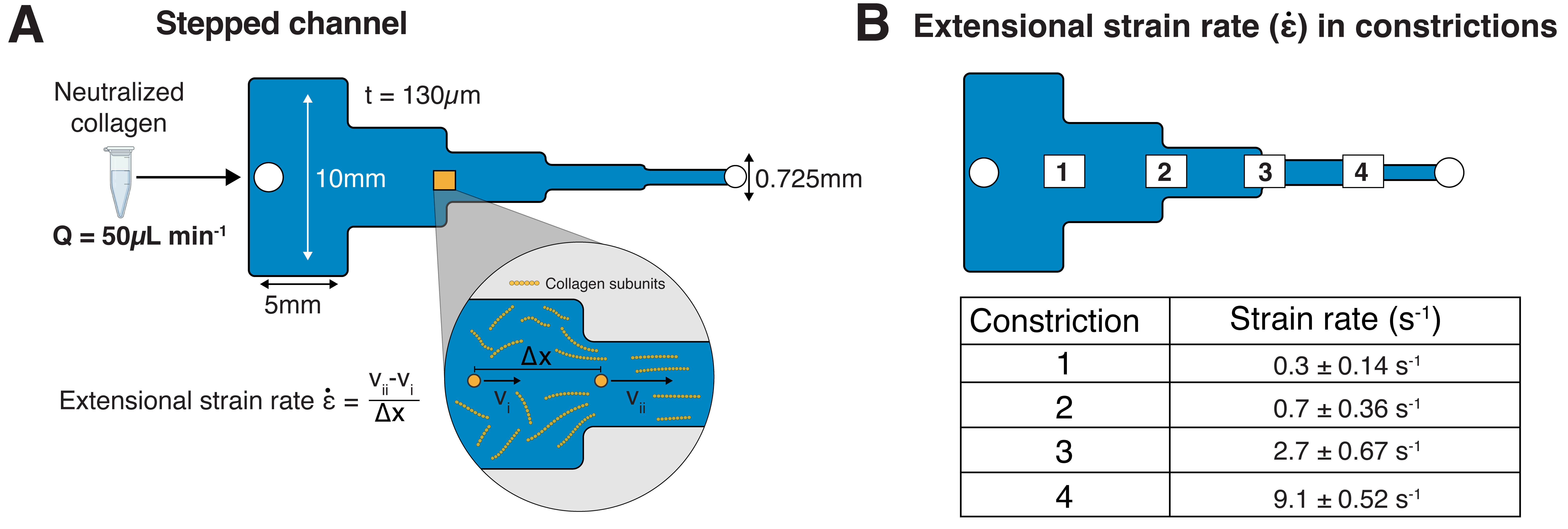
Extensional strain induced, long-range collagen fiber alignment in 3D hydrogels

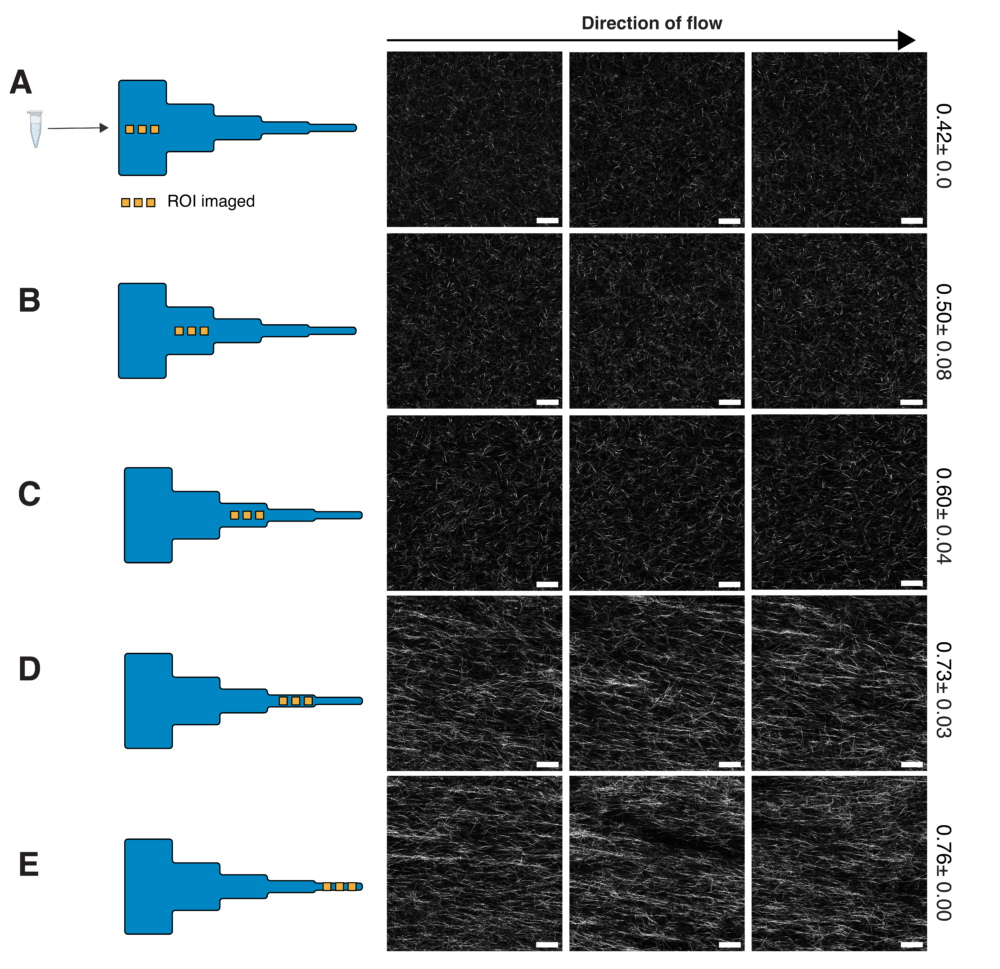
uniform channel. Panels (a-e) show representative images of collagen fibers in the corresponding
section of the segmented channel. The degree of alignment of collagen fibers is seen to increase
across the different segments. (B) Bar plot showing the mean COA ±SD in each segment, at a
flow rate of 50μL min-1. The COA was found to be greater than 0.5 at . (C) Bar plot showing
COA in zero strain conditions ( ̇ε =0) , and in combined extensional and shear flows ( ̇γ + ̇ε ).
The COA in the two conditions is not statistically different at lower shear rates and strain rates,
however, the mean COA increases rapidly at ̇ε = 2.7s−1. (D) representative images of collagen
fibers at a ̇γ =257s-1, in the zero-strain condition and at ̇ε =9.08s-1. Fiber anisotropy can be
visually observed to be higher with extensional strain. n=3. data represented as mean±SD. *
p<0.05, ** p<0.01, Scale bar = 25µm
Integration with SEAM for direct access to collagen
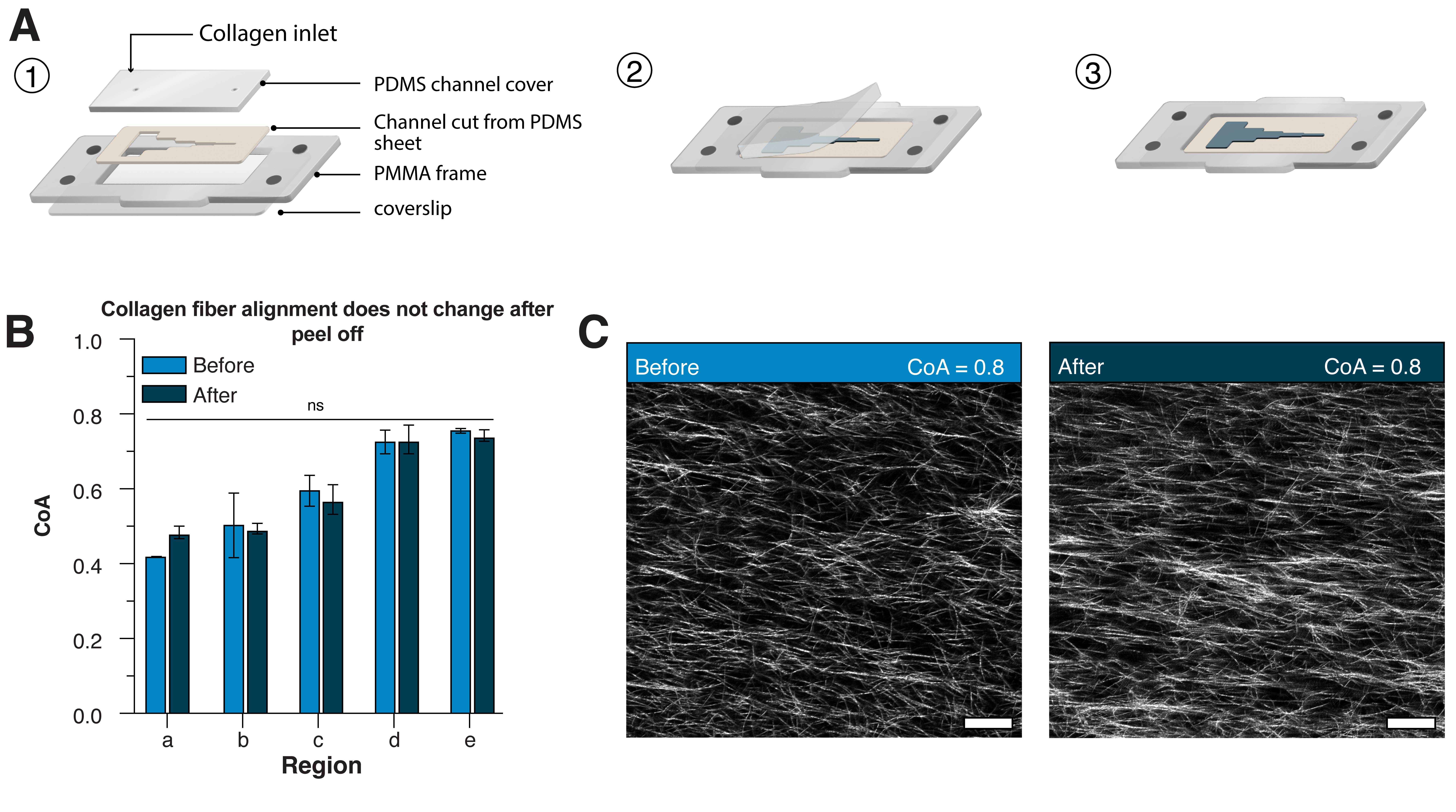
SEAM integration for cell culture and layer by layer fabrication