Exploring primary human microvascular endothelial cells for μSiM-hBBB modeling
Introduction
Sepsis is a maladaptive response to infection with a 25-50% mortality rate and a high incidence of cognitive decline among sepsis survivors.1-6 Because infection originates outside the brain, the factors responsible for cognitive decline in sepsis must get to the brain through the circulation and breach/act on the blood-brain barrier (BBB) to cause injury.7-9 It is impractical to directly image the damaged BBB of severely brain-injured patients, and mouse models of sepsis, while commonly used, do not give full response profiles resembling those of humans.10 Thus an in vitro model of the human BBB which includes circulating flow carrying potentially injurious factors, is necessary to study brain injury as an outcome of sepsis. Within this context, primary human microvascular endothelial cells (hBMECs) were initially considered as promising candidates due to the physiological relevance mirroring in vivo conditions. Our intention was to utilize hBMECs in μSiM-hBBB modeling to gain insights into the processes underlying brain injury during sepsis. However, our results showed that primary hBMECs may not be a good candidate for our μSiM-hBBB.
Results
During experimentation, primary hBMECs formed monolayers as expected and expressed junction proteins, i.e. Ve-Cadherin, ZO-1 (Figure 1). However, Claudin-5, a vital brain endothelial marker, displayed cytoplasmic localization instead of junctional distribution (Figure 1). Extending culture time did not improve the junctional Claudin-5 expression. Additionally, hBMEC monolayers exhibited high permeabilities (lucifer yellow, 3.31 ± 0.66 x 10-3 cm/min, Figure 2), which placed the hBMEC monolayers within the “leaky” or even “destructive barrier” category, contrary to our initial hypothesis.
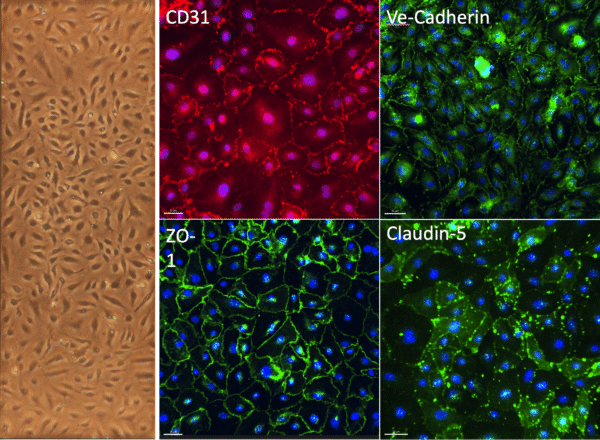
Figure 1 Phase-contrast image (4x), CD31, Ve-Cadherin, ZO-1, and Claudin-5 staining of p3 hBMECs. Cells cultured 6 days after confluency. Scale bar 50 μm.
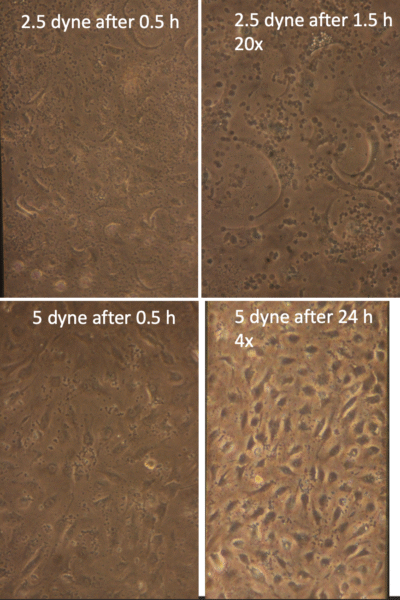
Figure 2 hBMECs got irritated under flow. Cells started losing under 2.5 dyne/cm2 after 1.5 h, but maintained under 5 dyne/cm2 after 24 h.
To enhance the barrier properties, I first implied shear conditioning to the hBMEC monolayer. Unexpectedly, hBMECs appeared irritated by the flow, leading to potential autophagy and the formation of vesicles within 0.5 hours. Interestingly, they survived 24 hours under a shear stress of 5 dyne/cm², but were washed away within 18 hours under 2.5 and 10 dyne/cm² (Figure 2). In pursuit of a solution, I attempted co-culturing hBMECs with normal human astrocytes (NHAs). However, this approach failed to resolve the issues of cytoplasmic claudin-5 and high permeabilities as well (Figure 3).
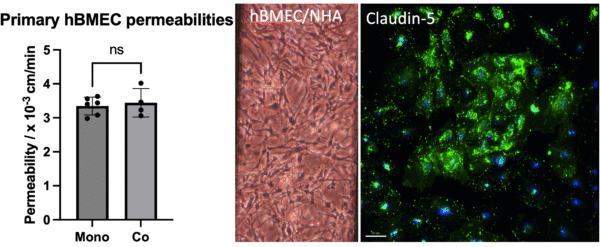
Figure 3 No significant change of permeability or Claudin-5 expression after cocultured with NHAs. hBMECs were cultured on the top of the membrane while NHAs were in the bottom channel. t test, ns p > 0.05.
Vendor Variability
Recognizing the potential influence of cell sources, I have explored different vendors for hBMECs. The hBMECs obtained from SciCell exhibited a transformation into fibroblast-like cells by passage 3, accompanied by a notable absence of CD31, an endothelial cell marker.
Our current primary hBMECs were derived from a 26-year-old female donor (motor accident). Despite the initial optimism surrounding the use of primary hBMECs for our μSiM-hNVU model, it is with great consideration that we must conclude that they do not meet the criteria we set for faithful BBB representation.
Conclusion
While primary hBMECs initially seemed promising for μSiM-hBBB modeling, my findings have led us down an unexpected path. Challenges such as the cytoplasmic localization of claudin-5, high permeability, and sensitivity to shear stress have raised questions about the suitability of hBMECs for my sepsis-focused BBB model.
In conclusion, the unexpected challenges encountered with primary hBMECs in my μSiM-hBBB modeling, we have made a strategic decision to transition to induced pluripotent stem cell (iPSC)-derived BMECs. Given iPSC-derived BMECs in our lab yielded promising results, particularly Claudin-5 junction expression and permeability, we believe that iPSC-derived BMECs will provide a more reliable and efficient platform for our studies, ultimately enhancing our understanding of this critical medical condition.
Reference
1 Singer, M. et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 315, 801-810, doi:10.1001/jama.2016.0287 (2016).
2 Iwashyna, T. J., Ely, E. W., Smith, D. M. & Langa, K. M. Long-term Cognitive Impairment and Functional Disability Among Survivors of Severe Sepsis. JAMA 304, 1787-1794, doi:10.1001/jama.2010.1553 (2010).
3 Murray, C. et al. Systemic inflammation induces acute working memory deficits in the primed brain: relevance for delirium. Neurobiol Aging 33, 603-616.e603, doi:10.1016/j.neurobiolaging.2010.04.002 (2012).
4 Annane, D. & Sharshar, T. Cognitive decline after sepsis. The Lancet Respiratory Medicine 3, 61-69, doi:10.1016/s2213-2600(14)70246-2 (2015).
5 Annane, D. & Sharshar, T. Cognitive decline after sepsis. Lancet Respir Med 3, 61-69, doi:10.1016/s2213-2600(14)70246-2 (2015).
6 Chavan, S. S. et al. HMGB1 mediates cognitive impairment in sepsis survivors. Mol Med 18, 930-937, doi:10.2119/molmed.2012.00195 (2012).
7 Opal, S. M. & van der Poll, T. Endothelial barrier dysfunction in septic shock. J Intern Med 277, 277-293, doi:10.1111/joim.12331 (2015).
8 Kuperberg, S. J. & Wadgaonkar, R. Sepsis-Associated Encephalopathy: The Blood–Brain Barrier and the Sphingolipid Rheostat. Frontiers in Immunology 8, doi:10.3389/fimmu.2017.00597 (2017).
9 Varatharaj, A. & Galea, I. The blood-brain barrier in systemic inflammation. Brain Behav Immun 60, 1-12, doi:10.1016/j.bbi.2016.03.010 (2017).
10 Seok, J. et al. Genomic responses in mouse models poorly mimic human inflammatory diseases. Proceedings of the National Academy of Sciences 110, 3507-3512, doi:10.1073/pnas.1222878110 (2013).