Small Particles Task Force: Initial Stock Preparation & Staining Workflow
Introduction
The LOMP Small Particle Taskforce is focused on developing a reliable and rigorous procedure for analyzing small microplastics in environmental samples. Our primary aim is to ensure that the data we generate accurately reflects the true microplastic population present in each sample. Achieving this requires careful attention to representativeness, verification, and contamination control throughout the entire workflow.
Three core objectives guide this effort. First, we must confirm that each sample analyzed is truly representative of the broader environmental system we are studying. Second, it is essential to verify that the particles counted are indeed microplastics, supported by appropriate analytical methods. Third, minimizing contamination from collection to processing to analysis is crucial for ensuring confidence in the final results.
To date, we have made significant progress in reducing methodological contamination and have begun refining our staining approach. However, further work is needed to better remove environmental contaminants and to verify that the particles we measure accurately represent the true population. This next phase of development will involve benchmarking to strengthen both reliability and reproducibility.
To better align the analytical method across LOMP, the MMC arm of the Small Particles Taskforce made stocks that all groups were asked to analyze with their current methods. This post walks through the process of preparing stock and analytical methods by the groups.
MMC Stock Preparation
Three stocks were prepared, which were just pure water prepared to be contamination-free; the second sample was PE beads (47-53 μm diameter), and the third was a combination of particles fabricated by the MMC that were <20 μm in size. The water sample was made by collecting MilliQ water in a MilliQ pre-rinsed glass jar. The water was taken to the URnano cleanroom and syringe filtered 2x to further clean the water. 150 mL of this water was saved for control runs; each lab was given 30 mL of the stock, while the rest was used to prepare 30 mL of the PE bead and MMC spiked samples. The PE bead sample was made by adding 200 μL of a 1 mg/mL solution of the beads to 30 mL of filtered water. MMC mixed particle samples were prepared by adding 200 µL each of PS (14.4 mg/mL), Nylon 6,6 (57.7 mg/mL), and PET (3 mg/mL) stocks to 30 mL of filtered water, giving final concentrations of approximately 94.1, 377, and 19.6 µg/mL, respectively (total ≈ 491 µg/mL).
| Table I: MMC Stock Preparation for SPTF |
|||
|---|---|---|---|
| Stock | Polymer Composition | Preparation Concentrations | Polymer Concentration |
| Water | 2x Syringe Filtered MilliQ water (control) | 30 mL of MilliQ water was collected in a pre-rinsed glass jar and syringe-filtered twice in the URnano cleanroom | NA |
| PE Beads | PE bead suspension in water (47–53 µm beads) | 30 mL filtered water + 200 µL of a 1 mg/mL PE bead stock | ≈ 6.7 µg/mL PE in water |
| MMC Spiked Mix | Mixed plastic particle suspension (PS, Nylon 6,6, PET, <20 µm) | 30 mL filtered water + PS (14.4 mg/mL), Nylon 6,6 (57.7 mg/mL), and PET (3 mg/mL) | ≈ 0.491 mg/mL total polymer (491 µg/mL) |
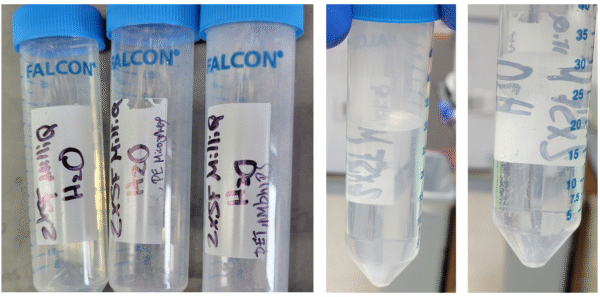 Figure 1: Prepared samples in 50 mL conical tubes.
Figure 1: Prepared samples in 50 mL conical tubes.
Nile Red Stain Preparation
| McGrath Lab | 20 µL of 1 µg/mL of Nile Red in 100% Ethanol |
| Robert Lab | Pre-stained samples with 5mL of 10 µg/mL Nile Red containing 0.01% Tween-20 |
| Elder Lab | 10 ug/mL NR in 100% Ethanol and filtered through a 0.5 μm Sepcon |