Porosity vs. Cell Morphology
As I was preparing for the BMES conference, I took a few pictures of endothelial cells on pnc-Si transwells that showed extensive vacuole/endosome expression (here). The most dramatic example occurred on a 50nm-thick membrane that was barely porous. So I wondered – are cells responding to mechanical or porosity differences (or both, or something else)?
To test this question, I assembled 15nm pnc-Si membrane (RTP’ed) transwells over a range of porosities. Specifically, I used SC303, SC264, SC324 and SC 348. All of these samples were pinhole-free in order to eliminate a pinhole’s contribution to porosity.
| Sample | Pore Size (%) | Porosity |
| SC348 | 0 | |
| SC264 | 0.198 | 6.2 |
| SC303 | 2.07 | 19.2 |
| SC324 | 5.24 | 23 |
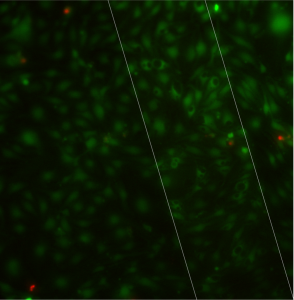
For this experiment, I seeded bEnd3 at 50000 cells/cm2 on the pnc-Si side and grew the cells upside down. I stained with Live/Dead after 7 days. I didn’t track TEER with these in order to minimize handling side effects. Here are the images in ascending order of porosity (free-standing area outlined in white):
SC348:
SC264:
SC303:
SC324:
There are vacuoles in cells on all of the samples. Qualitatively, I would organize the samples by increasing numbers of vacuoles as: low porosity (264), no porosity (348), medium porosity (303) and high porosity (324). The high porosity sample had lots of swollen cells concentrated on the membrane – I’m not sure if these are senescent cells or if the cells were swelling and releasing from the surface during the time it took for me to image these samples (they were last). Since there are vacuoles on both 0% and >0% porosity samples, one might conclude that this cell morphology does not depend on porosity. However, could the sample we label 0% porosity by TEM actually contain through pores that we missed in TEM, or contain pores that we assign as blocked but are actually open?
I also took a couple of images that I wanted to share:
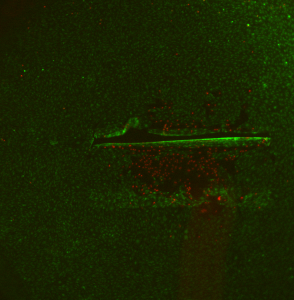
This shows that when a membrane breaks with a confluent layers of cells already present, cell death spreads far from the broken membrane. It would be bad to assume that cells on an intact membrane next to a broken membrane are healthy:
Here’s a 40X image of SC264, which shows a complex network of vacuoles. To get this image, I carefully placed the pnc-Si transwell on a glass slide in order to eliminate the dead space underneath the Sepcon in the multiwell plate.



Porosity sensing may be a threshold phenomenon so that a little porosity is enough to induce vacuoles. Mechanics isn’t as sexy a theory for us, but how will we test it?
One effect that does seem to trend with increasing porosity is the contrast between the cells over the membrane and the cells over the silicon frame. Do you agree? This would mean that the extra fluorescence that we always see for cells over the membrane is because the cells are able to pick up the dye more effectively when they are over the membrane and not because of signal reduction by the solid silicon.
To test mechanics, we could get Young’s modulus data from AFM/Bulge testing and then do cell culture on the same membranes. But we would have to know how to change the mechanics considerably without changing porosity.
I think it’d be easier to make a conclusion about cell brightness over the membranes if I scaled the fluorescence intensity to the same value for each image (which I didn’t do for this post). I only stain cells from 1 side now to eliminate the possibility of leading cells from both sides of the membrane. This is a metabolism-sensitive dye, so it could be that the cells are metabolically active over the membrane window.
Well the membrane cells still have 2X as much surface area available for uptake even if they are not seeing 2x as much dye. Its a little tough to sort this out, but more metabolic activity over the membrane would be terrific to prove.