Adventures in Bioprinting
Objective
- Develop an easily mainpulated reproducible vasculogenesis assay using hydrogels laden with endothelial cells (‘bio-ink’) patterned using the technique of extrusion bioprinting
Bio-inks
- Develop a bio-ink with the following characteristics
- Cell viable: bio-ink must sustain cell life, allowing cells to manipulate and move through the hydrogel
- Vasculogenic: bio-ink must encourage (or at least not negatively affect) tubule formation
- Printable: bio-ink must be viscous enough to extrude as a contiguous filament with appropriate dimensions
We realized early on that some of these “necessary characteristics” conflicted with each other. For example, the bio-ink must be stiff enough to be extruded as a contiguous filament (printability) and yet soft enough to sustain cell life (cell viability) and allow tubule formation (cell viability).
Given our experience with pro-vasculogenic Geltrex and Dr. Wan’s lab’s experience with printable Agarose, we decided to experiment with composite bio-inks with varying concentrations of Geltrex and Agarose.
Bio-ink conditions:
- 100% Geltrex
- 15:85 2 wt% Agarose-Geltrex
- 50:50 2 wt% Agarose-Geltrex
- 50:50 3 wt% Agarose-Geltrex
Agarose preparation:
- Appropriate amounts of agar powder added to boiling deionized water on hot plate
- Temperature decreased, solution stirred every 15 minutes for two hours until solution was clear
- Agarose transferred to conical stube and stored overnight in incubator at 37 °C to allow solution to further homogenize
Experiments
The following experiments were performed to assess the feasibility of the Agarose-Geltrex composite bio-inks. It was important that we tested the composite bio-inks for all three of the necessary characteristics. Therefore, we performed cell viability and vasculogenesis assays, as well as extrusion printing trails.
Cell Viability and Vasculogenic Capabilities:
Geltrex Control Preparation & Vasculogenesis Assay
100% Geltrex controls were executed to determine how the HUVEC remodeled the matrix, seeded either on top of the hydrogel or mixed in the hydrogel as a bio-ink.
For HUVEC seeded on top, 100% Geltrex was spread on the bottom of a 96-well and incubated at 37C for 1 hour to form a 500µm thick gel. HUVEC were seeded on top of the gel, and then flooded with Complete Media.
For the 100% Geltrex-HUVEC bio-ink, HUVEC were mixed into Geltrex to a concentration of 4% of the total bio-ink solution. The bio-ink was hand-extruded from a syringe into a 96-well and incubated at 37C for 30 minutes before being flooded with Complete Media. Half of the wells were also supplemented with 10 ng/mL VEGF.
Phase contrast images were acquired every hour to monitor tubule formation and ultimately vasculogenesis.
Results: HUVEC Capable of Vasculogenic Tubule Formation on 100% Geltrex Hydrogels
Vasculogenesis is defined as the de novo formation of capillaries and blood vessels from endothelial cells that ultimately form a primitive vascular network. Qualitative assays identify the occurrence of vasculogenesis through the observation of endothelial cell matrix remodeling and tubule formation. To confirm the ability of HUVEC to undergo vasculogenesis, HUVEC were seeded on top of 100% Geltrex hydrogels. Phase contrast images taken hourly confirmed the HUVECs ability to remodel the matrix and move across the gel. Early tubule formation was observed for the first 6 hours of culture. After 24 hours, the HUVEC developed a primitive vascular network as they formed connected tubules arranged in the typical honeycomb pattern. A Live/Dead assay at 24 hours confirmed that the HUVEC composing these tubules were alive. Media supplementation with 10 ng/mL VEGF did not affect the formation of tubules.
Composite Bio-ink Cell Viability & Vasculogenesis Assay
Cell Viability & Vasculogenesis bio-ink preparation methods:
For HUVEC seeded on top of hydrogels,
- Hydrogels were combined appropriately in chilled conical tubes and then transferred to a 96-well (35µL) using a pipette
- Hydrogels were incubated at 37C for 30 minutes
- HUVEC were added at varying seeding densities: 15,000 cells/well (recommended), 45,000 cells/well, and 90,000 cells/well, in 35µL of Complete HUVEC media
For HUVEC seeded within composite bio-inks,
- HUVEC were added at varying seeding densities: 15,000 cells/well (recommended), 45,000 cells/well, and 90,000 cells/well, to Geltrex in chilled conical tubes and gently mixed
- Agarose was quickly added to the cell-Geltrex solution and gently mixed
- Bio-ink was transferred to a 96-well and incubated at 37C for 30 minutes
- Wells were gently flooded with 35µL of Complete HUVEC media
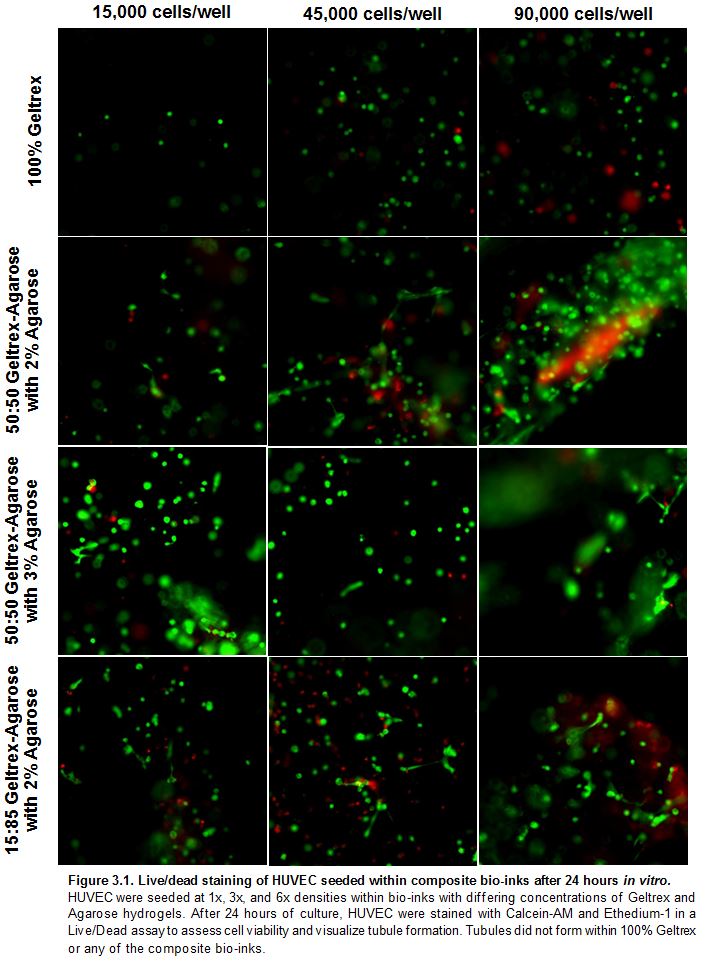
Results: HUVEC were able to survive within, yet unable to remodel composite bio-inks
HUVEC embedded within composite bio-inks were imaged hourly using Phase Contrast to monitor matrix remodeling and tubule formation. HUVEC were unable to remodel and move within the composite bio-inks, and therefore tubule formation did not occur. Even as seeding density was increased to 3x and 6x the original seeding densities, no tubule formation could be observed of HUVEC within 100% Geltrex or on top of or within the composite bio-inks. A Live/Dead assay confirmed that the cells that did not move were indeed still alive within the hydrogels.
Results: HUVEC Viability Decreased and Variable within Composite Bio-inks
Initially, we hypothesized that HUVEC cultured within 100% Geltrex would have the highest viability due to its growth factors and low stiffness that would allow the HUVEC to move. 50:50 Geltrex-3 wt% Agarose and 15:85 Geltrex- 2 wt% Agarose have comparable stiffness. We hypothesized that HUVEC cultured within these combinations would have the lowest viability because they are the stiffest bio-inks, and therefore not easily manipulated by the HUVEC. After 24 hours of culture within the bio-inks, HUVEC viability was assessed via a Live/Dead assay. Viability was quantified for each condition as the number of live cells stained green divided by the total number of live cells stained green and dead cells stained red. Viability was highly variable within each bio-ink. Viability between each bio-ink was not significantly different (Fig. 4A). Average percent viability in 100% Geltrex was the lowest of all the composite bio-inks, opposite of our original hypothesis. An explanation for this unexpected result may involve the state of the bio-ink during extrusion. When the 100% Geltrex bio-ink was being extruded into the 96-well, the bio-ink was still in a liquid state. The shear placed on the HUVEC while being extruded from the needle may have harmed a portion of the cells, decreasing overall viability. Data included in the appendix also exhibits the same trend of decreased viability of HUVEC extruded into the 96-wells immediately after mixing the bio-ink. HUVEC viability increases after partial gelation of the bio-ink and remains relatively steady during printing; therefore allowing time to pass between bio-ink mixing and extrusion does not adversely affect HUVEC viability. Previous studies have shown that the presence of VEGF induces HUVEC polarization and influences how HUVEC interact with the surrounding matrix. The addition of 10 ng/mL VEGF to the culture did not significantly affect HUVEC viability within each composite bio-ink (Fig. 4 B).
Printability:
Extrusion printing bio-ink preparation methods:
- HUVEC between P3 and P5 were passaged and reconsistituted using HUVEC Complete Media to a density range of 3,225 cells/µL to 12,550 cells/µL (media and cell solution constituted 4% of total bio-ink solution)
- Agarose solution was transferred to a 50 mL conical tube using a pipette
- HUVEC were added to the agarose and stirred using a pipette tip to homogenize
- Geltrex was added to the cell-agarose solution using
- 27G needle was attached to the printer syringe before composite bio-ink was poured in
- Plunger was inserted into the top of the syringe and pushed down to expel excess air
- Syringe was loaded into the bioprinter
Each material combination was extruded as droplets in 96-well plates, and as lines in both 6-well plates and 60x15mm petri dishes. The plates were placed on the stage and the printer was adjusted so that the syringe tip was located at the correct starting position inside the well/dish. Using the Seraph Robotics software, the extrusion rate was controlled at the printer’s maximum speed of 0.05 mm/s (Fig. 1). Post-printing, media was added to samples and the plates were stored in the incubator at 37 °C.
Results: Composite Bio-ink Printability & Qualitative Rheology
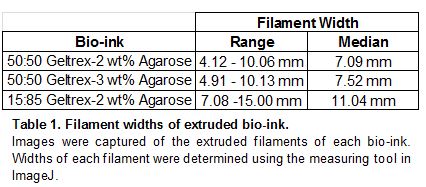
The ideal bio-ink would display high viscosity and stiffness in order to extrude contiguous filaments no wider than 500 µm to induce vasculogenesis in HUVEC. Quantitative rheological measurements could not be collected, therefore filament width measurements served as an indirect way to assess mechanical properties that would affect printability. Initial experiments were performed to determine the printability of 100% Geltrex. When extruded from a needle, this 100% Geltrex bio-ink acts as a liquid and spreads easily on a tissue culture surface. The 100% Geltrex solutions could be considered liquid droplets rather than contiguous filaments. Agarose of various weight percent and concentrations were added to the Geltrex to increase the viscosity and stiffness, and therefore improve the printability of the bio-ink by decreasing filament resolution. Filament width varied greatly within each composite bio-ink. Both of the 50:50 Geltrex-Agarose composite bio-inks with 2 wt% and 3 wt% Agarose had similar median filament widths of 7.09 mm and 7.52 mm, respectively. 15:85 Geltrex- 2 wt% Agarose had a larger median filament with of 11.04mm (Table 1). While the composite bio-inks had a significantly decreased filament width when compared to the 100% Geltrex bio-ink, none of the composite bio-inks came close to the target filament width of 500 µm.
Images of printing samples:
|
15:85 2 wt% Agarose-Geltrex |
|
50:50 2 wt% Geltrex-Agarose |
|
50:50 3 wt% Geltrex-Agarose |
Conclusions
This project aimed to develop a composite bio-ink that could be extruded into a micro-pattern to support endothelial cell tube formation. A vasculogenesis assay was performed to confirm the ability of HUVEC to form tubules on 100% Geltrex. HUVEC were able to form tubules with and without the addition of VEGF. Extrusion attempts illustrated that 100% Geltrex was not printable. The following composite bio-inks were tested to increase printability while maintaining cell viability and vasculogenesis potential: 50:50 Geltrex-2 wt% Agarose, 50:50 Geltrex- 3 wt% Agarose, and 15:85 Geltrex- 2 wt% Agarose. Timelapse imaging indicated that HUVEC were unable to remodel or move within any of the composite bio-inks. Live/Dead staining assays indicated highly variable cell viabilities within each material, but were not significantly different between the materials. The addition of VEGF did not significantly impact resulting HUVEC viability or observed remodeling capabilities. Filament widths of extruded gels were an order of magnitude greater than the target width of 500µm.
Although cells were able to survive in the composite bio-inks, none of the materials supported vasculogenesis or achieved target printability. Therefore, the composite bio-inks did not meet the criteria of a material that could be used for a reproducible, bio-printed vasculogenesis assay. Future experiments should explore different hydrogel combinations to develop an ideal composite bio-ink that is biocompatible with HUVEC, facilitates vasculogenesis, and can be reproducibly extruded with appropriate dimensions.