w685 diffusion
I attempted a diffusion study with w685 three days ago. If you were following the comments on the w685 page, you’ll know I had some difficulty pipetting the protein solution into the well. I haven’t had this problem recently, and when I used to encounter this problem I would etch the remaining backside oxide with 4:1 HF.
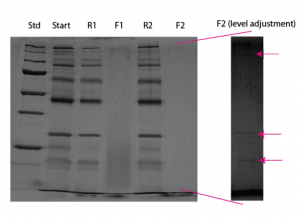
The results from the gel I ran yesterday for this experiment had me thinking that I did not actually get any protein in contact with the membrane because I saw no bands in the filtrate. This may not be entirely true as I see a couple of ghost bands in F2 if I mess with the pixel intensities. I’m not sure why only these bands are seen and why so faintly.
Today I went to retry this assay with all intention of etching the oxide with HF and then setting up the experiment as usual. However, the HF balled up on the test chip whereas it should have spread if the chip was indeed hydrophilic. I tested another chip with water, and the I verified the chips were now more hydrophobic. What could cause this temporal difference in hydrophobicity? I guess this increase in contact angle is similar to what I observed in my contact angle studies, but it seems the changes in this case have taken place over a longer time period.
I set up two new 685 chips with protein standards, and will post diffusion results here once I obtain them.
Additionally, there was a minute amount of discoloration during these 24 hour experiments. There are patches of purple on these chips, which started out a lighter blue color.
I set up the next batch of 24 hour diffusion tests with w685. As I mentioned previously, these chips already had a hydrophobic top surface. Gel results are on their way soon. Here’s an image of the chips after the experiment was finished:
One discolored more than the other, but not enough to compromise the membrane in 24 hours. However, I found it intriguing that the second chip had some very high spacial discoloration (arrow). This chip is from location -4,2 of an underetched wafer, meaning that this chip was not totally perforated and brought along a little bit of the outside wafer region along with it when I popped it out. This region apparently discolors faster. It is thinner than the bulk wafer because while it is partially but not fully etched down to this surface. Does this mean attack from the backside??
Gel Results:
Unfortunately this turned out to be a cruddy gel. The tube with one of the retentates burst in the heat block, and I lost all of that sample. I ran the remaining samples on the gel and obtained results similar to the previous experiment that I thought was “broken.” The first filtrate shows a couple of lower bands, but the actual passage rate was low. The second filtrate shows no passage at all. Both positions were r=2.8.
Maybe my first experiment wasn’t a bust after all. Even though this wafer has small pores, I’ve never seen so little passage of protein before. Pressurized results will be coming soon, but just as a spoiler, I noticed very low (non-existant) flow rates.