Visualization of Endothelial Focal Adhesion Formation on Transparent Porous SiO2 Membranes
Introduction
Permeable membranes provide a physiologically relevant cell culture substrate for in vitro study of barrier cell types, such as endothelial cells, which assume a polarized morphology with distinct apical and basolateral surfaces. Cell morphology and subsequent monolayer formation and barrier function are largely dependent on initial interactions with the cell culture substrate via focal adhesions. Previous studies have shown that cells fully embedded within a 3D matrix will not form focal adhesions, while cell cultured on 2D substrates will form focal adhesions. We hypothesize that a highly permeable, ultrathin membrane could act like a “slice” of a 3D matrix. We tested this hypothesis by investigating the formation of focal adhesions on optically transparent SiO2 membranes with 0.5 µm pore sizes and 19.6% porosity (high porosity, HP).
Methods
The SiO2 membranes were fabricated using standard MEMS processing techniques. 300 nm of SiO2 was deposited using PECVD and annealed to create a tensile film on polished silicon wafers. Both device geometry and pore structure were patterned using common photolithography techniques. Pores were formed using a reactive ion etch. Membranes were fabricated with 0.5 µm and 3.0 µm pore sizes with 19.6% (HP) porosity.

Fig. 1. Comparison of Membrane Pore Size
Figure 1 shows a 0.5 µm HP SiO2 membrane and a 3.0 µm HP SiO2 membrane.
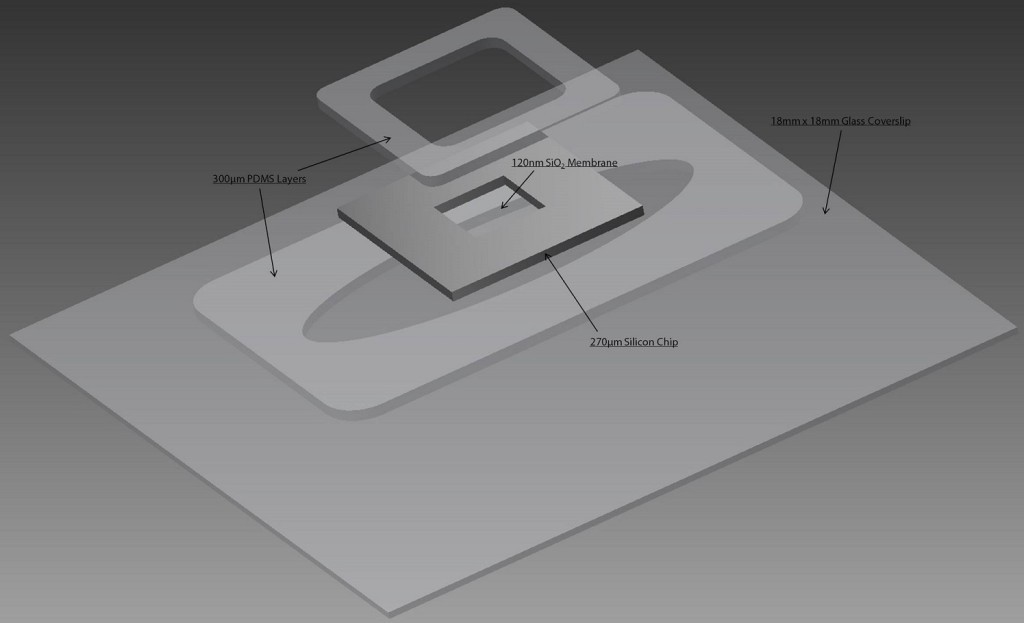
During the fabrication of the cell culture devices, SiO2 membranes were bonded to custom cut silicone gaskets and then to the bottom of 24-well plates using ozone bonding, and UV sterilized. For analysis on commercial tissue culture plastic (TCP) substrates, a single silicone gasket was bonded to the bottom of a well. All SiO2 membranes and TCP substrates were pretreated with a 1% Geltrex coating.
 ]
]
Fig. 2. Single-Well CytoVu Model
The 3D model shows how the single-well CytoVu holds the SiO2 membranes in place using 600 µm silicone gaskets bonded to TCP. The trench cut in the center of the lower gasket allows for media to flow both above and below the membrane for cells to receive nutrients.
Human Umbilical Vein Endothelial Cells (HUVECs) were cultured on porous SiO2 membranes, as well as non-porous SiO2 membranes and TCP for 2D controls. Cells were permeabilized with Triton X-200 detergent and then fixed. To visualize nuclei, F-actin, and vinculin, cells were stained using DAPI, phalloidin, and anti-vinculin antibody, respectively. To visualize nuclei, VE-cadherin, and vinculin, cells were stained using DAPI, anti-human CD144, and anti-vinculin antibody. Cells were imaged via fluorescence microscopy using a dry 40x objective on a wide-field microscope. Metamoprh and ImageJ software were used image analysis.
Visualization of Focal Adhesions
Focal adhesions are large macromolecular assemblies through which mechanical forces are transmitted between a cell and then extra-cellular matrix. Previous studies have shown that focal adhesions form on the basal surface of cells grown on stiff, non-compliant 2D culture substrates. Focal adhesion formation occurs on these substrates in response to the relatively large amount of force exerted on the cells by the substrate. Similar studies have shown that focal adhesions are not readily detected in cells grown on soft 2D substrates or cells fully embedded within a 3D matrix. Focal adhesion formation does not occur won these substrates due to the relatively low amount of force exerted on the cells by the substrate.
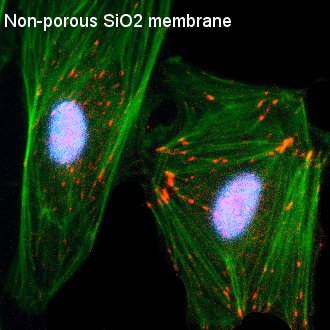
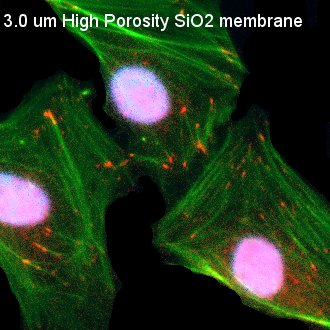
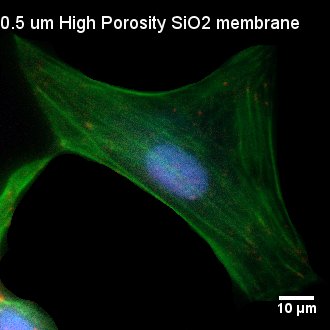
Fig. 3. Visualization of Focal Adhesion on Various Substrates
HUVECs were seeded on TCP, as well as non-porous (NP) SiO2, 3.0 µm SiO2, and 0.5 µm SiO2 membranes. Cells were permeabilized, fixed, and stained 24 hours post-seeding. Cells were stained with DAPI (nuclei, blue), phalloidin (actin, green), and anti-vinculin antibody (vinculin, red). Nuclei appear pinkish in some images due to bleed through int the red fluorescence channel during imaging. TCP and NP SiO2 membranes served as 2D controls with contiguous surfaces for comparison with porous SiO2 membranes. Comparable, punctate focal adhesions were readily observed on both TCP and NP SIO2 membranes. The size, shape, and general conformation fo the focal adhesions on the NP SiO2 membranes appeared similar to focal adhesion formation on different substrates with OH-based surface chemistries investigated in different studies. Focal adhesions became less distinct on 3.0 µm SiO2 membranes and were scarcely observed on 0.5 µm SiO2 membranes.
Quantitative Analysis of Vinculin
Vinculin is a structural molecules that is concentrated on the cytoplasmic side of focal adhesions. This linker protein serves as a mechanical stabilizer by attaching F-actin to the plasma membrane and binding to other linker proteins within a focal adhesion, such as talin and paxillin. The quantification of vinculin, and subsequently the quantification of focal adhesion formation, is a method of assaying the extent of cell attachment and perception of the surface to which it is attached.

Table 1. Quantitative Analysisof Focal Adhesion Formation on Various Substrates
The number of punctate focal adhesions were manually counted using an ImageJ plugin and are displayed in the table above as the average number of focal adhesions per cell plus or minus the standard error of the mean. The number of focal adhesions per 1000 µm² of cell spread area are presented in the table above as the number of focal adhesions per cell spread area plus or minus the standard error of the mean. Previous studies have shown that proliferation rates and cell spread area are comparable between the substrates. The number of punctate focal adhesions were nearly identical between TCP and NP SiO2 membranes. This value for the 3.0 µm HP SiO2 was lower and significantly different from TCP and NP SiO2 as indicated by p<0.05. This value for the 0.5 µ HP SiO2 was even lower and significantly different from TCP and NP SiO2 as indicated by p<0.01. THese results demonstate that as the amount of contiguous surface area on a substrate decreases, the formation of focal adhesions after 24 hours also decreases. HUVEC respond to the NP SiO2 membrane more like a 2D substrate and to the 0.5 µm SiO2 membrane more like a 3D substrate, while the 3.0µm SiO2 membrane falls somewhere in between.
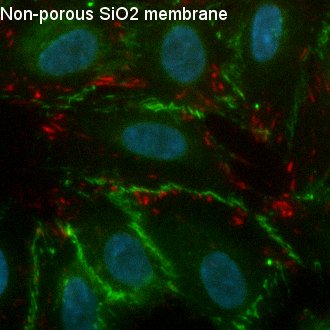

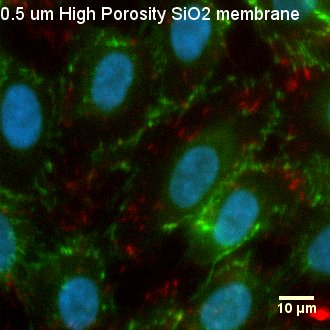
Fig. 4. Visualization of Focal Adhesions and Cell-Cell Junctions
After 6 days of culture, HUVEC were permeabilized, fixed, and stained with DAPI (nuclei, blue), anti-human CD-144 (VE-cadherin, green), and anti-vinculin antibody (vinculin, red). Cells on all three substrates formed a monolayer, expressing the junctional molecule VE-cadherin at their cell borders. Cells exhibit both unstable, remodeling junctions demonstrated by the finger-like VE-cadherin protrusions and stable, mature junctions displayed by the zipper-like VE0cadherin structures between cells. Cell-cell junctions and barrier formation are important in endothelial cells as these characteristics affect the formation of blood vessels cells.
Conclusions and Summary:
- Purpose of experiment: How do cells behave on physiologically relevant, ultrathin, optically transparent, permeable SiO2 membrane compared to TCP?
- Cells on 2D substrates form focal adhesions while cells in 3D matrices do not due to differences in force signals
- Cells on TCP and NP SiO2 formed nearly identical number of focal adhesions per cell, therefore cells respond to NP SiO2 like a 2D substrate
- Cells on 0.5 µm and 3.0 µm SiO2 both formed a lower number of focal adhesions per cell and were statistically different from TCP and NP, therefore cells respond to 0.5 µm SiO2 like a 3D substrate and 3.0 µm SiO2 is intermediate
- As area between pores decreased, the formation of focal adhesions also decreased