HUVEC ECM Deposition, Porous Substrates, and The Feed Forward Loop That Leads to Cell Detachment Under Flow
Introduction
Endothelial cell detachment under flow on porous substrates has been a concern in our sepsis research since I began my work in the lab, and some time before that as well. As we plan on moving to microfluidic models that incorporate larger pore substrates and physiological shear stresses for extended periods of time, it is clear that adherence issues must be addressed before research can proceed. Most of my work on cell adherence to date has focused on morphological outcomes of cell culture on our materials (i.e. cell alignment and basic cell attachment or sustained culture under flow). To fully grasp this hindrance, we must first ask why we are presented with these issues from a biological stand point, and then work on addressing said issues.
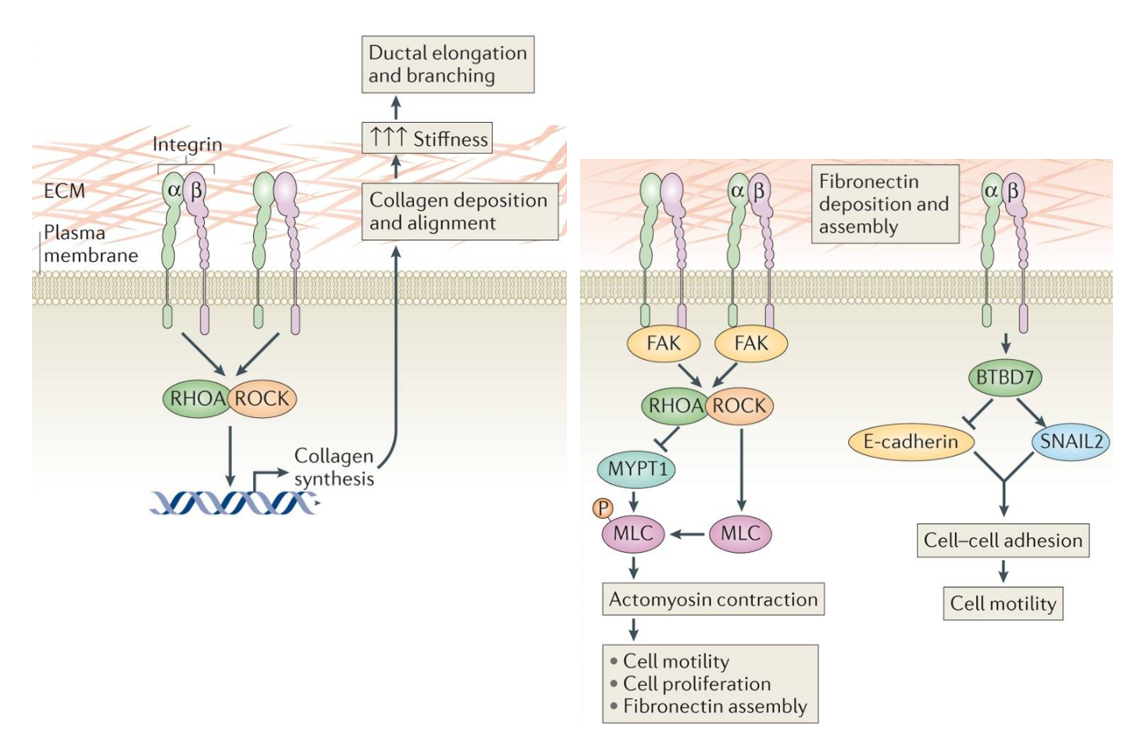
In order for cells to proliferate, they must bind to the substrate on which they are seeded. This concept is not novel by any means, and has been studied extensively to date. Cells form these cell-substrate adhesions through membrane associated dimers termed integrins. Integrins are made up of alpha and beta subunits that bind to a variety of ligands. In the case of endothelial cells, the important ligands in vivo are fibronectin, laminins 8 and 10, and collagen 4. These three components together form the basement membrane on which endothelial cells grow. When culturing cells in vitro, it is important to consider replication of the basement membrane to ensure healthy cell growth. It has also been shown that cells remodel and deposit their own ECM through integrin signaling (Figure 1) in a feed-forward mechanistic fashion. For these reasons, we have consistently functionalized our porous materials with human fibronectin (0.6 mg/ml) prior to cell seeding.
In all cases, HUVECs grow on our fibronectin functionalized porous membranes in static conditions, however, when introduced to flow (10 dyns/cm^2), cells on the microporous membrane shear off. While the issue seems very black and white (larger pores means less area for the cell to grip and thus cell detachment), the question becomes very interesting when porosity (void space/membrane area) is held constant. As seen previously, cells grown on 3 um membranes at 20% porosity shear off within hours of the introduction of flow, where as cells grown on 0.5 um membranes with the same porosity take longer to show detachment. This phenomena is seen again when we switch to the NPN materials (porosity varies, but is close to 15% from what I can find in most cases) where cells remain healthy for extended periods of time and align in the direction of flow. All together, this preliminary data tells us that while void space is still a major factor in cell adherence, ECM patterning can be just as important. This leads to another question though; can cells produce basement membrane fibers that span our pores, and how does increase in pore size effect this phenomena. I hypothesize that cells produce ECM in a time dependent manor that is strongly influence by pore sizes due to a decrease in focal adhesion formation on microporous substrates.
Methods (Proposed)
To see if our cells produce ECM fibers over our pores, I propose the use of SEM imaging. SEM of HUVEC produced ECM has been done in the past (Figure 2), however, the introduction of our membranes and varying time points is key.
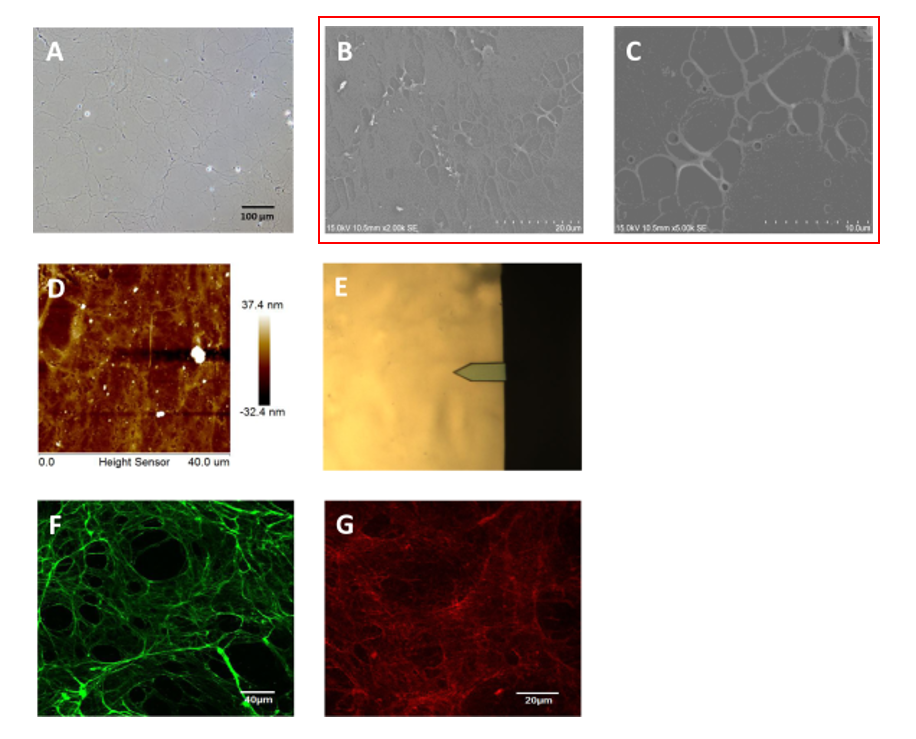
Following the given papers protocol, I plan on performing the following experiments:
Seed HUVECs on NPN and 3 um HP SiN at varying densities to produce confluent monolayers after 24, 48, and 96 hours.
This may take some trial and error, but doubling times in literature and experience to date will be used to calculate optimal seeding densities.
Culture Decellulatization
Confluent cells will be rinsed with PBS and chilled to 4 C before incubation with a solution containing 0.5% Triton X-100 and 20 mM NH4OH at 37 C for 5 mins.
Cells will be rinsed with PBS and treated with 100 units/mL PBS DNasel and incubated at 37 C for 45 mins.
SEM Fixation
HUVEC ECM will be washed with PBS and fixed in 22.5% glutaraldehyde for 1 hour.
Progressive dehydration with ascending ethanol concentrations will be performed, followed by critical point drying.
ECM will be coated with gold-platinum(?) and imaged under the SEM
Results and Disscusion
While I hypothesize cells will produce a uniform ECM on both substrates eventually, I do believe well will see the immediate influence of pore size on ECM deposition at the earlier time points. How this experiment pans out will drive my aim, so it is hard to propose a future experiment at this point, but I see it going one of two ways: either we see ECM formation over pores on the microporous substrate at the earlier time point, which would leave me to believe that cells are being influenced by patches of compliant substrate, causing them to detach, or ECM formation is highly substrate dependent, and the adherence issue is easily solved (although nothing is ever easy on these membranes) by increasing culture times. If the result is the former, Tom proposed the use of AFM to prob for ECM compliance over the pores to seek out a model for increased inter-spaced compliance and cell attachment, which may give us a good idea of how cells interact with their substrate in an spacial manor. One outcome I have yet to address, however, is the chance of ECM never spanning 3 um pores. While I believe this outcome is unlikely, it will mean we will have to address the adherence issues less from an integrin signaling stand point and instead try to solve the issue through the use of something cells bind to stronger than fibronectin (i.e. poly-l-lysin).
Alec, can you add the citations and links for the 2 papers you used for images?
Done.