Phalloidin Staining on RPE-19 cells
Some of my recent cell culture work on Microporous MgF2 windows have shown vacancies in the cultured monolayer after fixation and staining. We should see a contiguous layer. To better understand my fixation, I seeded and cultured P10 RPE-19 cells at a high density on a coverglass and in a polystyrene dish for a week. This is not long enough to generate tight junctions (ZO-1) in this cell line, but is long enough for cell confluence. I put a gasket on top of the coverglass to constrain the area of cell growth, much like in the devices I use.
Many thanks to Alec for helping me with the Phalloidin staining. I fixed with warmed 4% PFA in cell culture media (lacking FBS) for 30 minutes in the fume hood, and permeabilized with 0.1% Triton X-100 in PBS. I used DAPI at 1 ug/mL, and then stained with Phalloidin.
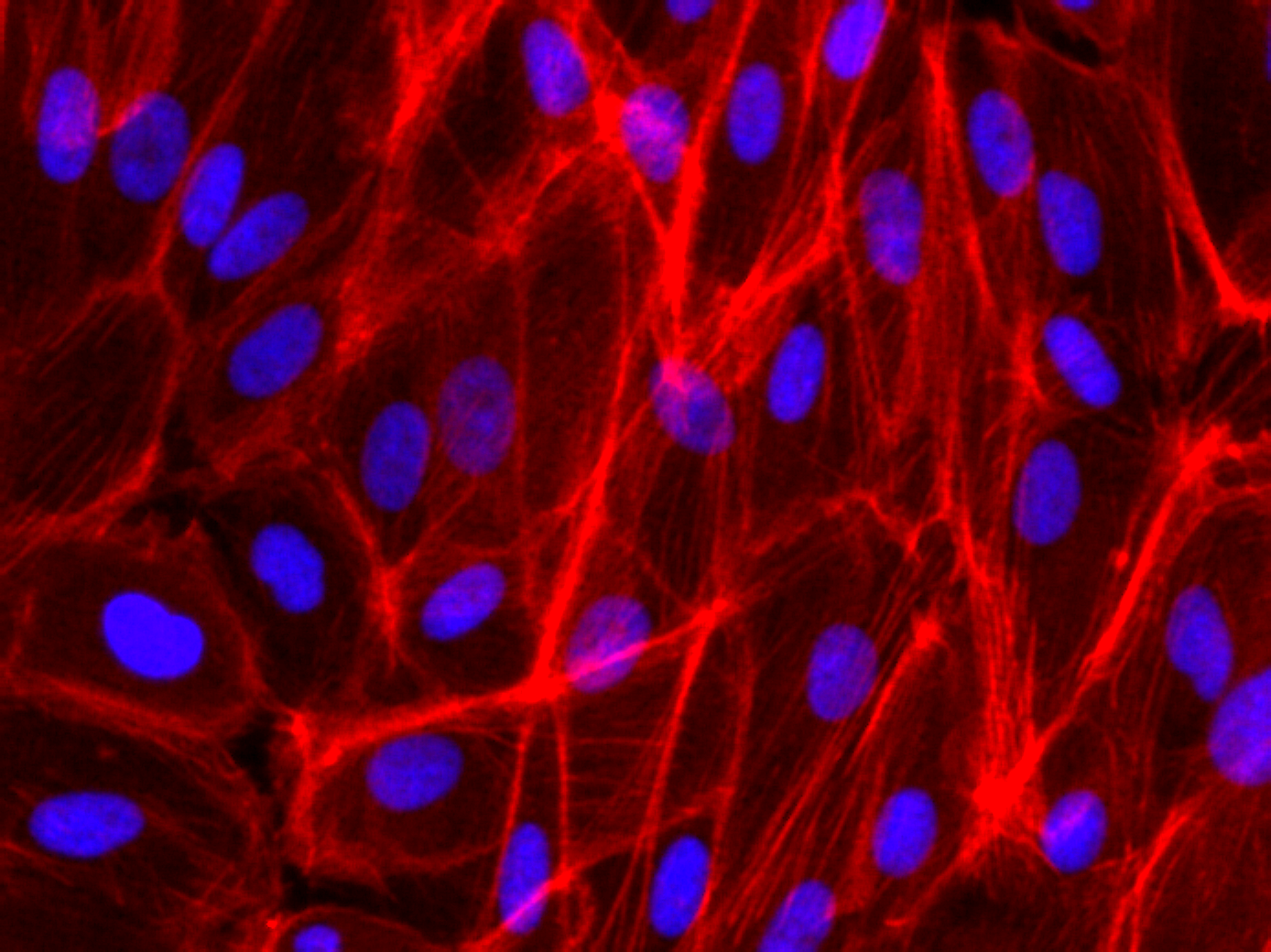
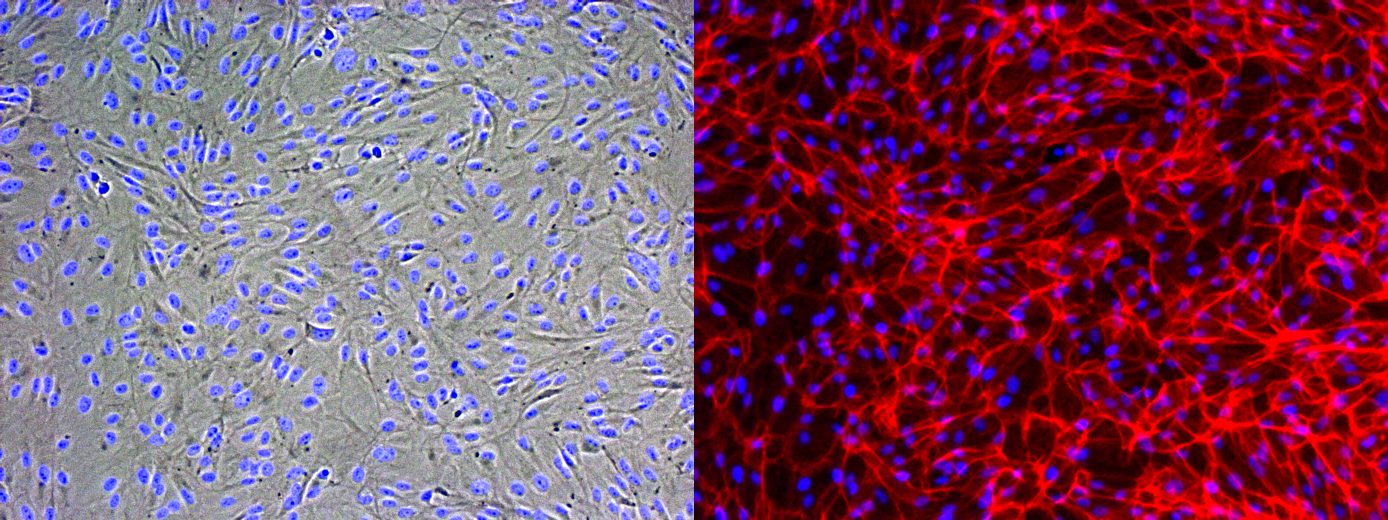
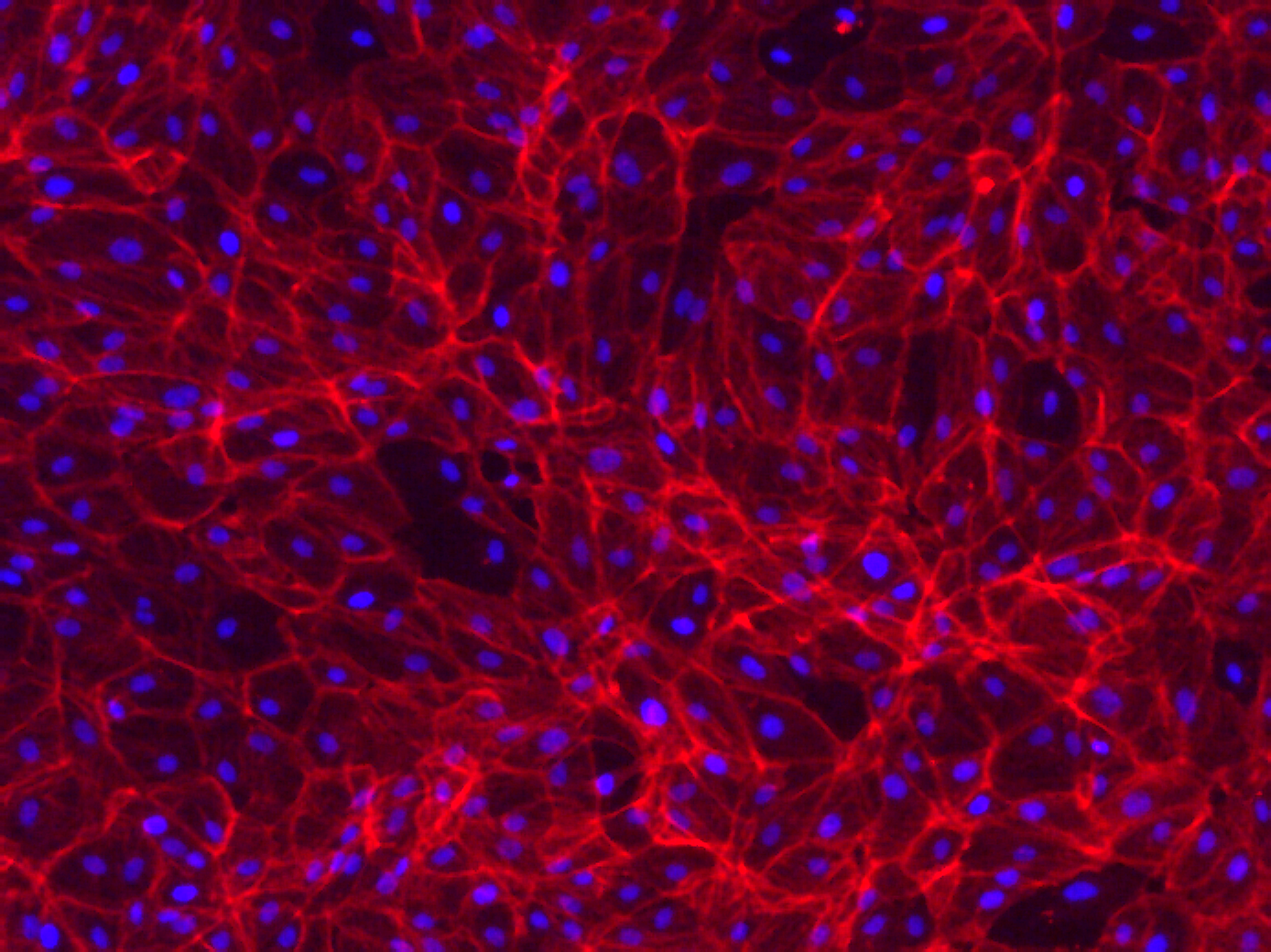
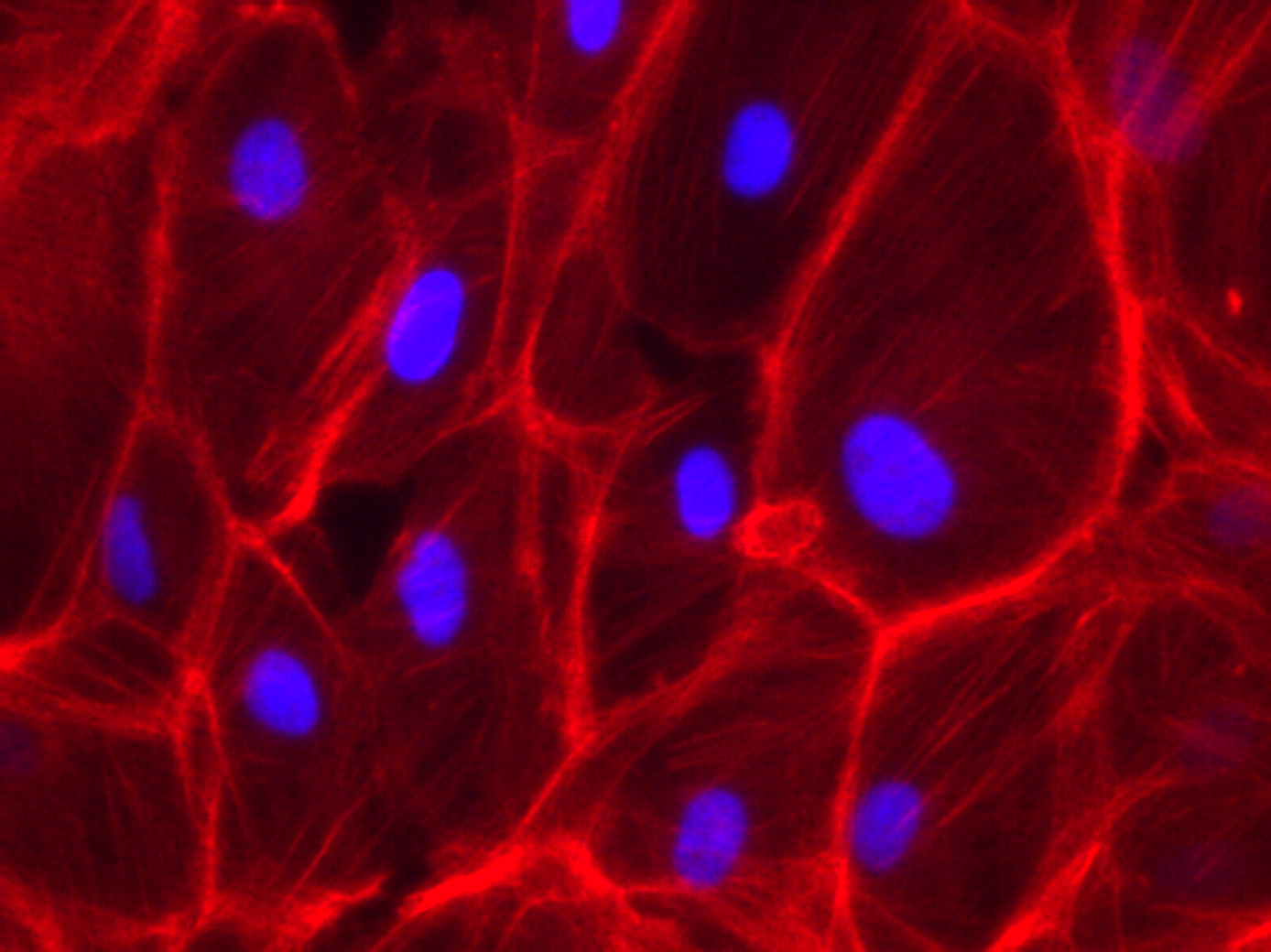
It’s possible you’re over-fixing the cells with 30 min in 4% PFA – 10 min should be more than sufficient especially if pre-warmed. Over-fixing can cause morphology and also desirable epitopes to suffer a bit.
What Kevin mentioned is more important when you go to antibody staining I believe, but is a good point. I asked my histology/microscopy instructor about ICC on our devices and she said 15 mins of 4% PFA fixation is probably not going to effect our imaging. I still think trying to incubate your cells for a longer time on a cover slip and then trying your ICC protocol again with lower fixation time might solve some of your issues.