hiPSC Differentiation to Endothelial Cell Culture Method, Brain Microvascular Endothelial Cells (EECM-BMEC-like cells), Round 2
Introduction
This post highlights results from my second attempt to differentiate hiPSCs into endothelial progenitor cells EECM-BMEC-like cells in our lab. For details on methodology, see Nishihara et al., FASEB, 2020 and my previous post.
Methods

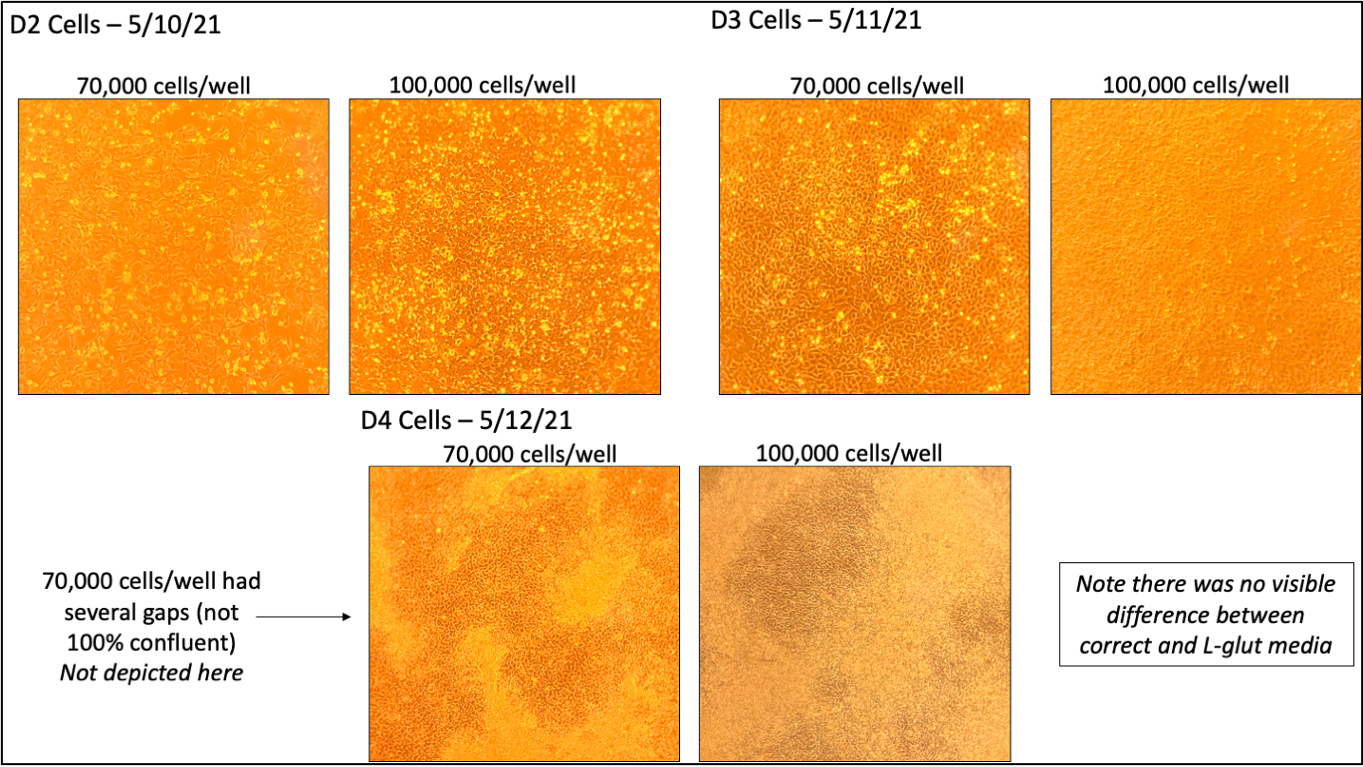
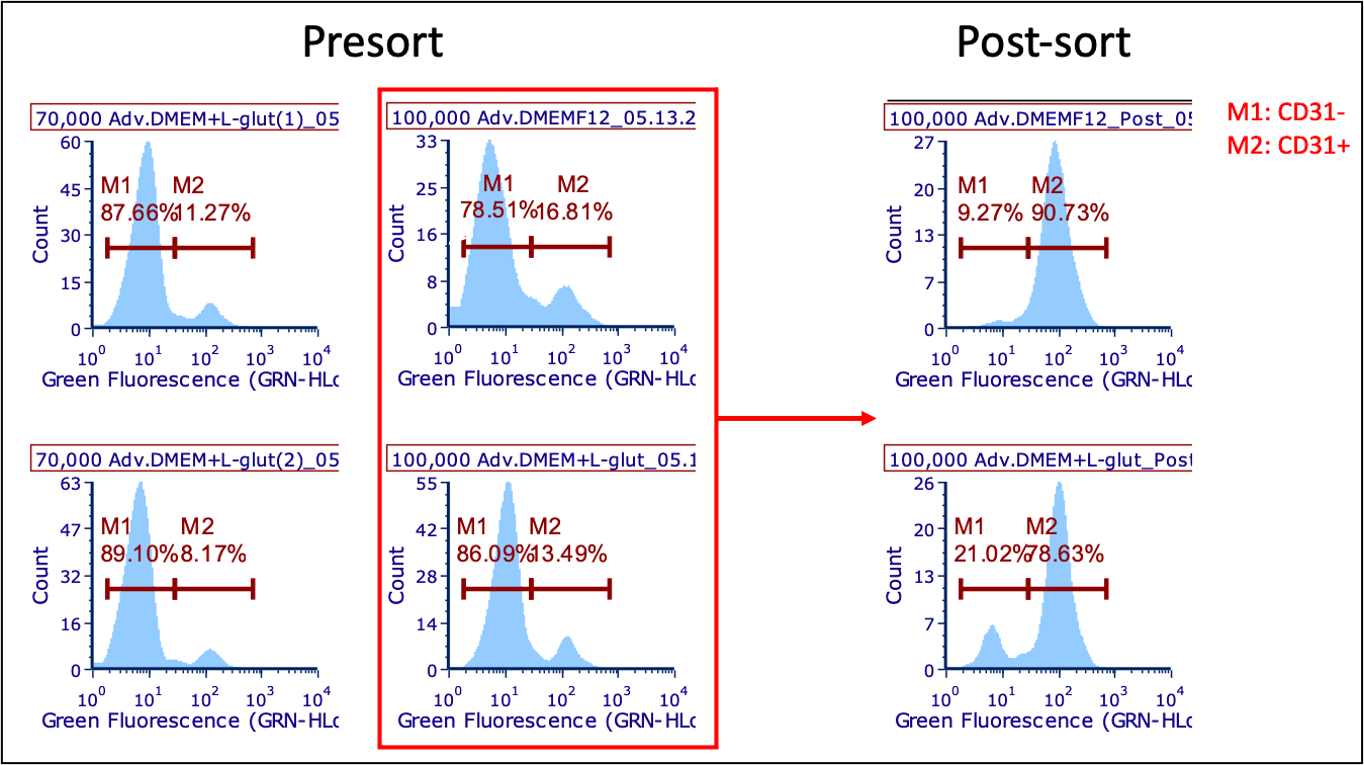
Cells were seeded at 70,000 and 100,000 cells/well on D-3. Only the 100,000 cell/well plate was sorted. Due to media being backordered, some cells were maintained in LaSR made with Advanced DMEM + Ham’s F12 (with L-glutamine), instead of Advanced DMEM/F12 (no L-glutamine). This cells were not used for any analyses. Following MACS, one well was plated at 300,000 cells/well and selectively passaged twice before using for analysis.
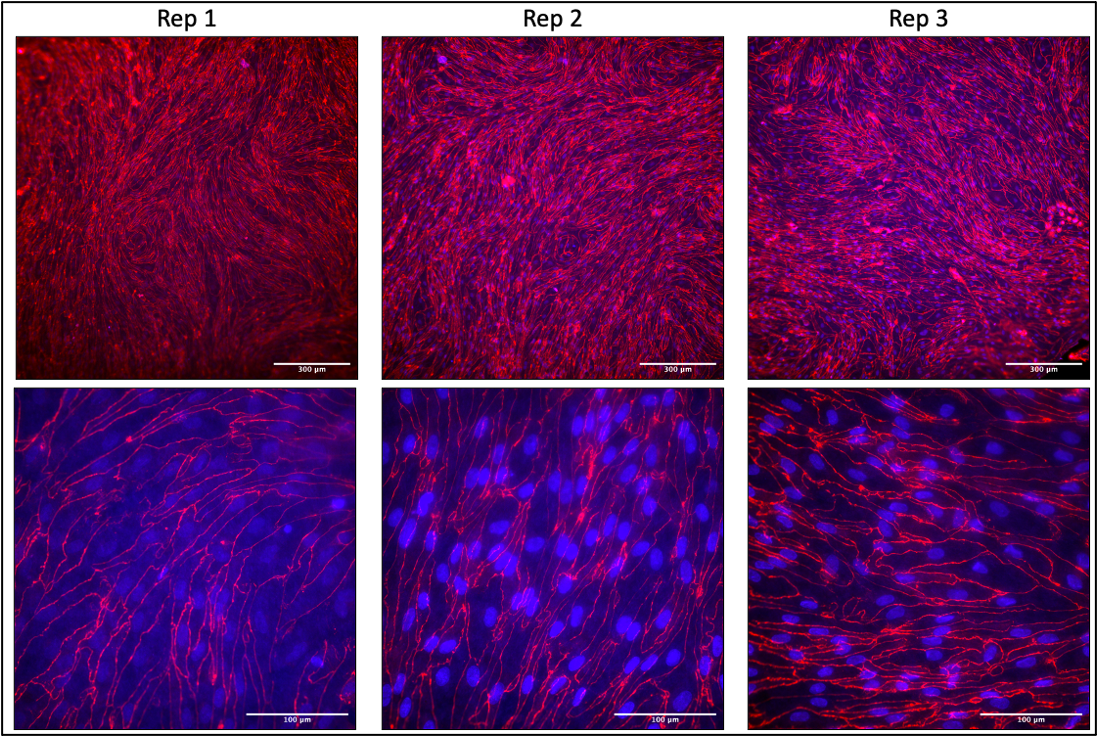
At passages 2-4, a subset of cells were seeded into collagen IV (400 μg/mL, dissolved in 0.5 mg/ml acetic acid)/fibronectin (Gibco, 100µg/ml)-coated 6.5mm diameter, 0.4 µm PC Transwell filters at a density of 100,000/cm2 in hECSR medium. TEER measurements were taken daily (EndOhm chamber using EVOM2 system), and permeability was measured after 6 days of culture. Transwell filters were fixed, removed, and stained for ZO-1 to check monolayer maturity. Note that cell growth slowed following passage 4.
At passage 3, a subset of cells were seeded into collagen IV (400 μg/mL, dissolved in 0.5 mg/ml acetic acid)/fibronectin (Gibco, 100µg/ml)-coated µSiMs with single slot NPN membranes (lot 1364) at a density of 15,000 cells/device in hECSR medium. Media was replaced 2 hr after seeding and once the following day. After 2 days of culture, cells were fixed and stained for VE-cadherin (R&D Systems), PECAM-1 (Thermo, PA5-32321), Claudin-5 (Thermo 35-2500), Occludin (Thermo 35-1500), and ZO-1 (Thermo 40-2200).
At passage 4, cells were seeded into a collagen IV (10 μg/mL)-coated 24 well plate at a density of 95,000 cells/well and cultured for 3 days. After two days of culture, hECSR was replaced with SMLC-CM with or without 1 ng/mL of recombinant human TNF-α (R&D Systems) and 20 IU/mL recombinant human IFN-γ (R&D Systems). Proinflammatory stimuli were maintained approximately 20 hours before cell adhesion molecules analyzed (Guava EasyCyte Flow Cytometer). THE PLATE WAS DROPPED DURING SECOND WASH, CAUSING SPILLOVER BETWEEN WELLS AND CELL LOSS.
During passages 3 and 4, 1-2 x 106 cells were resuspended in 1 ml freezing media (hECSR supplemented with 30% FBS, 10% DMSO, and 5 µM Y-27632) and frozen for future use.
Results


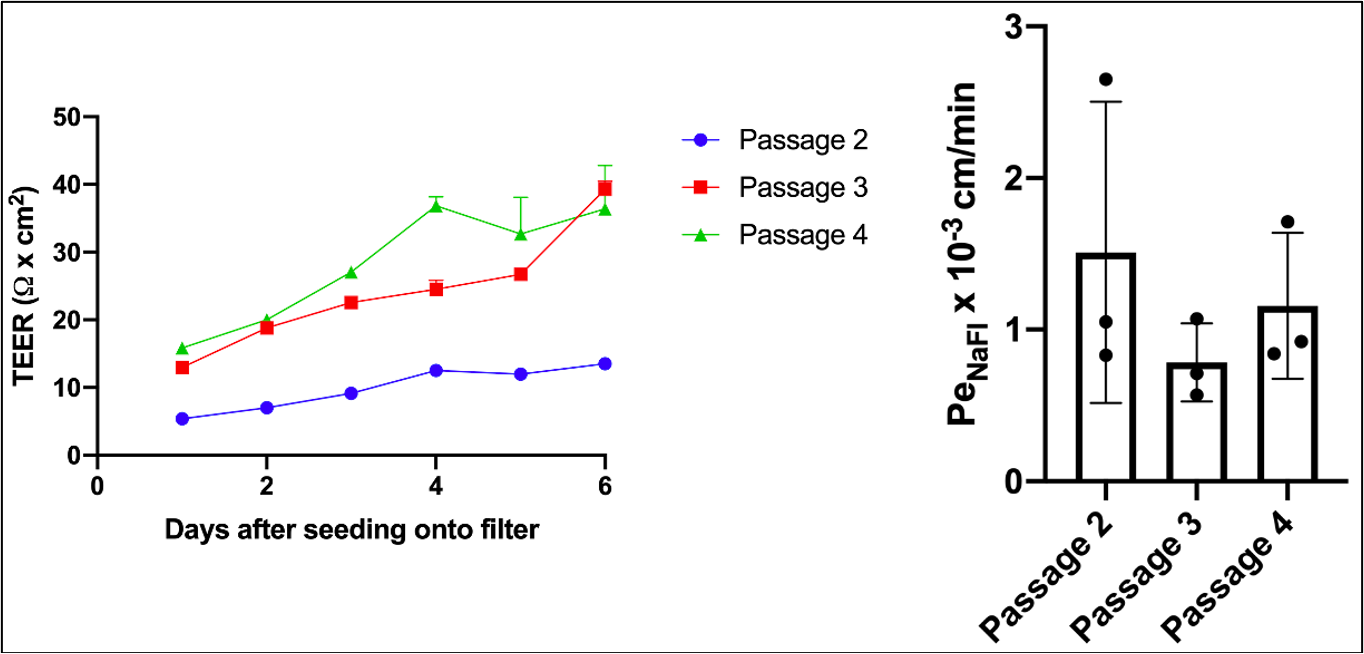
TEER was improved compared to the previous experiment, but still lower than reported in Nishihara et al. Permeability was near the published values obtained for IMR90-4 (~0.7 x 103 cm/min), but below what Hideaki has achieved with this cell line (≤0.5 x 103 cm/min).
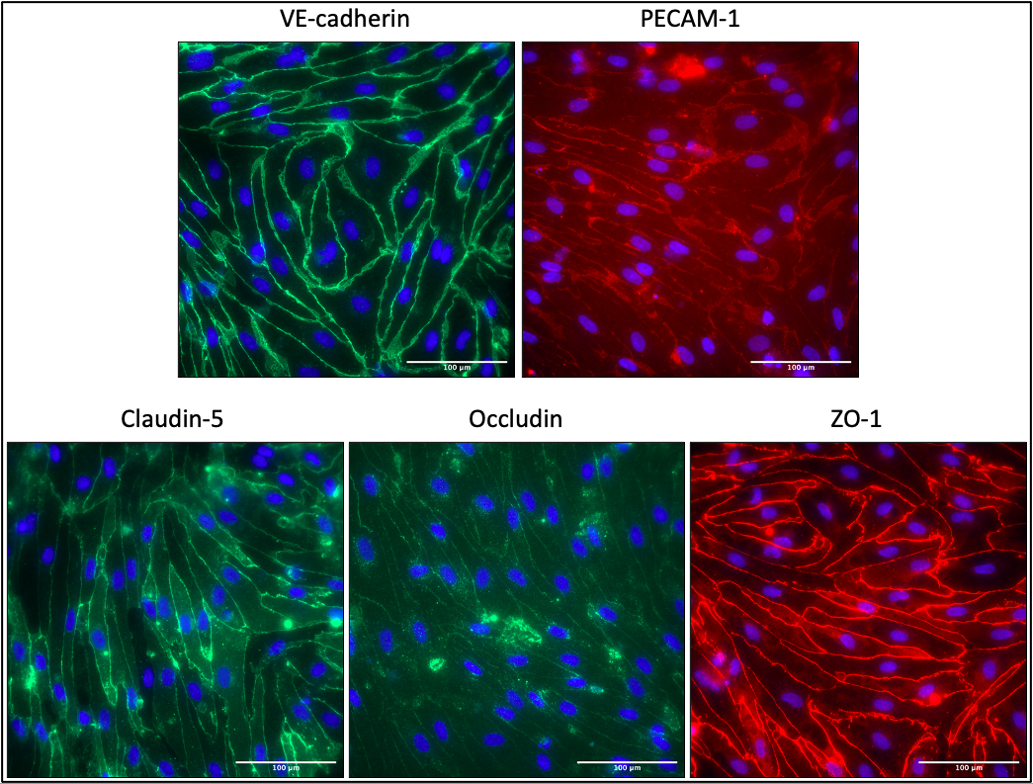
Cells express all junctional molecules, comparable to what is seen in Pelin’s post.
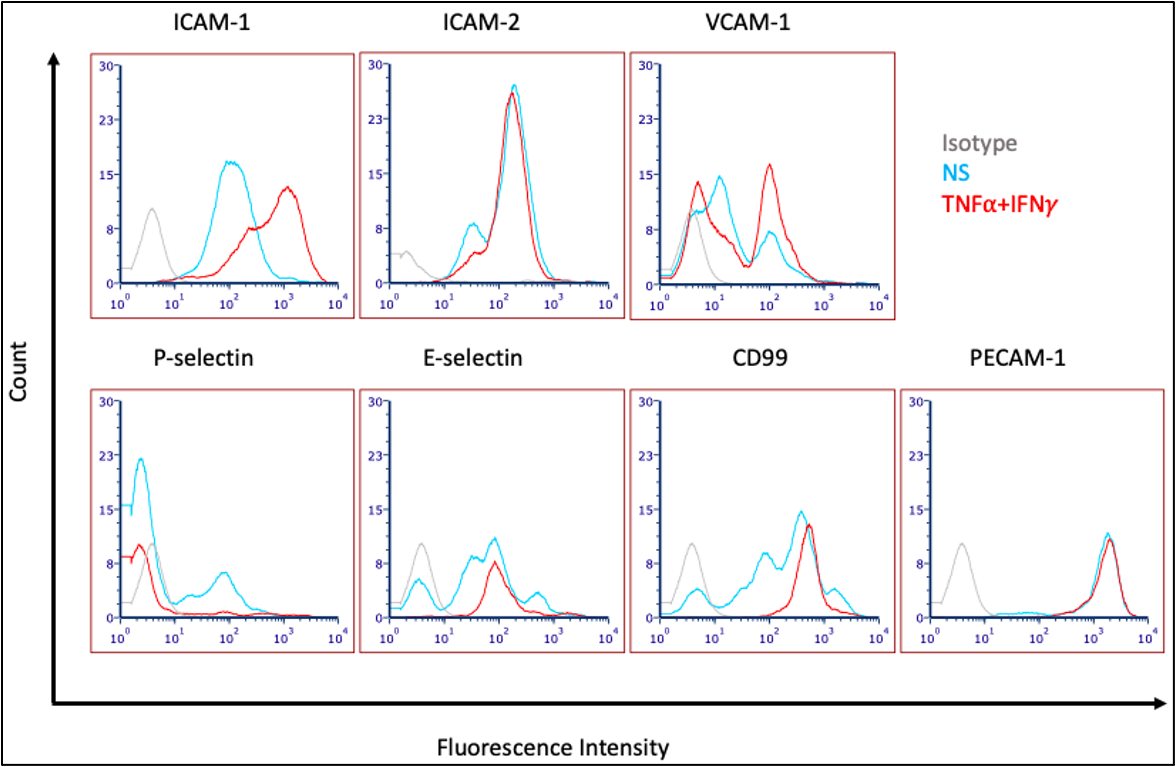
Data is hard to interpret due to the plate being dropped and wells contaminating each other. It is unclear whether we see VCAM-1 expression with culturing in SMLC-CM and unclear if we have expression of selectins, measured with the new antibodies. Data is suggestive that we at least have E-selectin expression now, and possibly VCAM-1 and P-selectin expression, but this experiment will need to be repeated.
Conclusions
This second round of EECM-BMEC-like cell differentiation was an improvement from the first round, with better expression of junctional markers and lower permeabilities/higher TEER. Next experiment, I will not do TEER and, instead, culture cells on 12 well format Transwell filters rather than 24 well format filters to see if surface area makes a difference on permeability. Further, purer EECM-BMEC populations were obtained, and two vials of EECM-BMECs were frozen for future use. These vials will be used to repeat flow cytometry of cell adhesion molecules following proinflammatory stimulation, in addition to ICC on cell adhesion molecules following proinflammatory stimulation of cells cultured in µSiMs.