EO rate changing over time
I have observed that in some cases the rate of EO can go up or down over the course of an experiment. Often the initial burst in the first 5 minutes is high, then the rate may stabilize or drop. I have observed that the rate of EO seems closely related to the current. Here is an elongated experiment in which the rate increased much more dramatically than I was expecting (w170, no ox).
The initial rate jumped after the first 30 minutes. The final rate is almost 9 ul/min, way higher than the old oxidized membranes. The membrane appeared intact at the end of the experiment. I confirmed that a broken small square membrane does not have any flow due to EO.
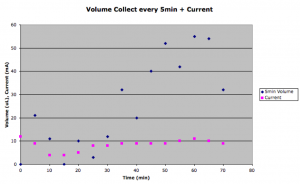
Here is a breakdown of the raw collected volume and current at each 5 min time point.
Initially there’s a big jump in volume passed (possibly due to bubble development), and then the current and the amount collected drops. The current then increases to the beginning value, and the amount collected jumps even higher than the initial burst.
I’m not sure how to explain this. Maybe there’s a wetting phenomenon at play.

How much is the temperature changing in your experiments, as I see this as another variable that could be changing over these very long time periods? This could then affect the wetting or surface charge distribution, or have some other effect.
This reminds me of the work that Shigeru has done on our membranes. He had considerable difficulty fully wetting our membranes in his SECM measurements. This is why I have always felt that we may not be wetting our membranes completely, so have room for improvement if we eventually figure this out. I could see wetting as another effect that could happen over this time scale.
If you keep the membranes wet, will this new rate be stable indefinitely? Once wet, I see no obvious reason why they would de-wet, whereas surface charge effects could continue to vary if the system is allowed to sit for some time to return to thermal equilibrium.
I checked with Shigeru, and in order to get uniform SECM images of the membrane surface, he had to ozone clean them, then soak the membranes in IPA for >3 minutes and then displace the IPA with electrolyte without drying. I think this is the only direct data that we have to evaluate our wetting properties, correct? If so, I tend to believe it. Therefore, it seems reasonable that the surface wetting is a probable theory.
I’m soaking a couple membranes in IPA overnight. I’ll try running an experiment until it settles, turn off the field, and then rerun after some time also.