EO with Ag/AgCl electrodes on membrane chip
I tried putting Ag/AgCl electrodes on the surfaces of the membrane for EO pumping. The experiments were done in RIT with Jirachai.
In these experiments the goal was to observe the change in liquid droplet volume on the front side of the membrane, due to EO pumping from liquid reservoir on the other side of membrane. To prepare the samples, I first seal the edges of membrane chip in silicone so it can float. Then I attach silver wires on both sides. Next, silver chloride ink is painted on top of attached silver wires. During experiment the silicone sealed sample floats in container filled with 0.1M NaCl, which allows the backside of the membrane and AgCl back electrode to be immersed in liquid. A droplet of NaCl of known volume placed on top surface of the sample partially covers membrane slits and top electrode. The droplet volume change due to EO is later observed in microscope when voltage is applied.
Prepared samples:
Below are two videos showing that droplet volume changes when 1V and 2V are applied. I sped up these two videos (x 4):
In next video the polarity of electrodes was changed every 20s so the droplet is pumped up and down. In this video you can see that the electrode changes color from silver to black few times. This is due to dissolution of AgCl. The video is sped up (x 8):
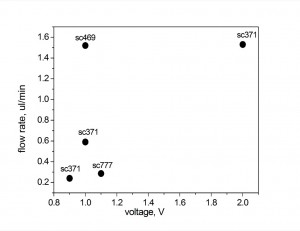
I calculated the flow rates from the videos assuming that the droplet contact angle doesn’t change. The flow rates vs applied voltages for different membranes are below. Wafers in the graph have different membrane areas (sc371-2 slits, others 5 slits).
The numbers are similar to what Jess got with Ag/AgCl electrodes. The flow is not constant, it slows down with time. Also the flow rate in forward direction (droplet pumping up) is higher than in reverse. The major problem in such setup (and in thin film Ag/AgCl electrodes in general) is quick dissolution of AgCl which influences the flow.
At some point during experiment the top electrode starts to degrade and dissolve into solution. Not sure why it happens, may be due to degradation of resin mixed with AgCl in the ink or its poor adhesion to silver epoxy. This degradation is shown in the video below:
Let me know if you have problems viewing videos in the post.