Fibroblast Growth: Viability and Discoloration
I recently attempted a fibroblast (L1 3T3 cell line) growth test. The cells were seeded at too high of a density, so growth curves can’t be produced. However I checked cell viability and pnc-Si discoloration.
As expected RTP treated samples discolored later than treated samples. Samples with cells seemed to discolor sooner than samples without cells; I will monitor this for the next few growth experiments to see if this is a real trend.
Cell viability was very similar between glass, plastic, and pnc-Si. However, this may not be the real story as culture media removal and washing may remove floating dead cells.
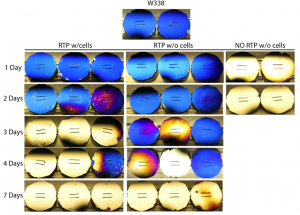

I’m concerned about the effectiveness of the RTP treatment. Are the samples with cells treated any differently then samples without cells. Is the media replenished more frequently, for example? What fraction of the surface was covered with cells in these experiments? An estimate will do. Was it 20%, 50%, 80%?
The RTP treated samples with and without cells are treated the same. The samples were RTPed two days before the experiment began. They were sterilized with methanol, washed with media (EGM), coated with gelatin, and then again placed in media. I changed the media every 2 days in both cases. The RTP samples were removed daily for imaging, including washing with deionized water and drying with 70% ethanol. The samples without cells were reused, whereas the samples with cells were different for each time point. I would estimate the cell density to be 40% on day 1, 75% on day 2, and confluent from then on.
We started growth tests with HUVEC and L1 3T3 growth tests yesterday using samples from W510 that were RTPed the same day as the experiment was started. We will monitor discoloration to see what happens.