Desalting BSA: Blank pnc-Si and Mini-Dialysis Units
The ultimate experimental goal of the desalting project is the removal of salts from a salt/protein solution while fractionating certain sized proteins with maximum protein retention and recovery.Thus far we have characterized the flux of salt through our membranes which show flux an order of magnitude greater than cellulose membranes. Our next step is to dialyze a protein/salt solution, measuring flux and protein retention over time.
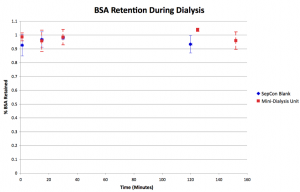
In this experiment 1 mg/mL BSA in 1M NaCl was dialyzed for ~2 hours in mini-dialysis units (cellulose, 10,000 MWCO) and blank (no membrane slits) SepCons. Conductivity of the deionized water surrounding was monitored over time. Samples were taken from the SepCons/mini units at 1, 15, 30, and 120 minutes to measure the amount of retained BSA. The Bradford Assay using a standard curve created from various concentrations of BSA in deionized water was used to quantify the amount of protein retained.
As expected, protein retention was very high for the blank SepCon as it has no pores and should act as a sealed container; this is supported by an insignificant change in conductivity. The mini-dialysis units also showed high retention, as BSA should not pass through a membrane with a 10,000 molecular weight cutoff. Furthermore, retention stayed constant for the duration of the experiment, suggesting the protein is retained while the salt is removed. Deviation from 100% retention may be due to variation in the Bradford assay, loss of protein due to absorption to the membrane/pnc-Si/plastic, or handling.
I should also mention that flux of salt through the mini-dialysis unit was the same for 1M NaCl with and without BSA, 2E-6 M/(mm^2*min).