Negative Adsorption Assay
This is a retrial of the negative adsorption assay using the Tecan spec/fluorimeter that we have been demo-ing. To perform this assay, I am applying a known concentration to the surface I want to test. After an hour I remove some of the solution and measuring the remaining concentration using absorption and the Bradford assay. This should give us a clue on how much of the protein is leaving the solution and sticking to the material.
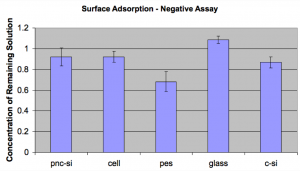
First Trial: Starting concentration is 1mg/mL BSA. Here is a chart of the results:
From this it looks like PES adsorbs the most protein. Glass for some reason appears to have more than the starting amount of protein, and I was concerned that maybe some evaporation was going on here. I decided to do another trial and I wanted to lower the starting concentration to see if I would get a more visible effect.
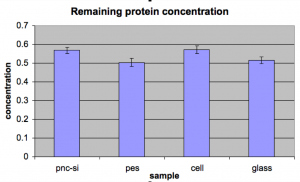
Second Trial: Starting concentration was .5mg/mL BSA. Here are the results:
To my chagrin, all of the sample were above the starting concentration. I looked back at my standards, and due to the line fit, the .5mg/mL standard could be back calculated as .55mg/mL. This is part of the discrepancy, but it doesn’t explain how pnc-Si got up to .57mg/mL. This time I was also as careful as I could be about evaporation; all experiments were done in sealed humidity chambers.
I should keep in mind though, that the pnc-si, pes, and cellulose are following the same trend in these two charts. Both pnc-Si and cellulose are adsorbing very little while pes is adsorbing more. Although the fact that glass is not similar across the two charts may be enough to throw out this observation too.
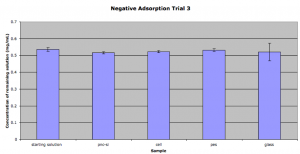
Here’s the third trial of this setup. I’ve included a bar that shows the concentration of the starting solution after the calculation from the standards:
This time all of the results fall more or less within the noise. There is no longer the trend of PES being more adsorbing either. Again I’m a bit at a loss of how to proceed with this study.
Write to this guy: Bharatkumar Bhut. bbhut@clemson.edu and ask him some questions about how he does this. This is the guy I met at GC. He does this at a different scale, but I doubt his assay is inherently more sensitive. I still say the secret to making this work is in the proper ratio of protein/volume/substrate, so you need to figure out what these are for him and adjust everything proportionately down to your dimensions.