Permeability causes vacuole formation
Another one of the questions I’ve been trying to answer with regard to endothelial cell vacuoles is whether membrane mechanics or permeability drive vacuole expression. I’ve thought of several experimental designs to test this that haven’t worked (i.e., using very small volumes on the apical side to minimize permeability, using PDMS plugs to block the well-side). I’ve finally settled on a system that should work to test whether permeability drives vacuole expression (critiques welcome). It’s sort of a pseudo-transwell set up:
The bottom cloning ring is attached to the pnc-Si with vacuum grease. There is also a ring of grease on the inside of the bottom cloning ring to prevent media from spilling out of the ring. After cell adhesion, another cloning ring is attached to the stop of pnc-Si with grease, or a sterile glass cover slip is placed over the wells. Therefore, the left set-up mimics a transwell with media on both sides of the cells and the right set-up limits the media on one side to the well volume only. Cells grow on the bottom, on the pnc-Si side.
I seeded the normal 50000 cells/cm2 (P9) and allowed them to attach for 2 hours. I then arranged these set-ups and allowed the cells to grow overnight before staining in Live/Dead solution.
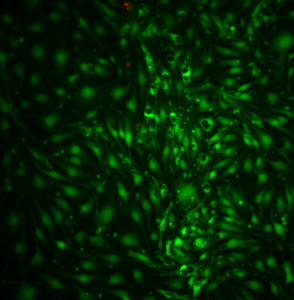
The left set-up yielded cells with numerous vacuoles. This suggests that this set-up mimics the Sepcon transwells decently well:
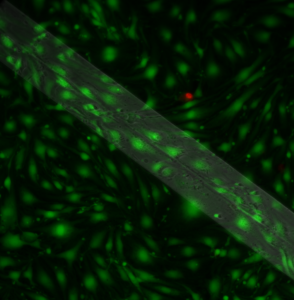
The right set-up yielded cells with no vacuoles over the membrane:
Unfortunately, I lost the second sample in this set-up due to membrane breakage. Also, it looks like this membrane is split down the middle. However, these results strongly suggest that permeability drives vacuole formation!

Awesome! A very good day for this project!
Lets get some more images so we can choose the prettiest pictures for publication. Can we keep these cultures going and confirm that we don’t see tubules?
Now the fun part: connecting it to biology. What have we learned about endothelial cell function? Is there any literature that this story fits into? Our ability to put this into a cool context will determine whether this makes a big splash or just gets published as some interesting phenomenon. Can we make this the result the positive answer to the test of an interesting hypothesis, preferably one that others already care about. True this is not how the practice of Science is taught in the textbook, but it’ll do.
Clever set-up.
Is there any fluid in between the cover slip and the membrane in the configuration on the right? I’m a little concerned that in the process of cutting off the permeability, a new structure was created with air bubbles/interface within the membrane pores. Any idea if this would affect cell growth?
I still think that absolute confirmation requires a silicon membrane without pores, but this experiment is getting very close…
thanks JP.
Chris – for the set-up on the right, I “pre-wet” the well side and then aspirated off most of the media. Then I carefully placed a coverslip over the wells. The media spreads out across the surface and I’m pretty sure that the well volumes remain full of media. I share your concerns about an air/liquid interface which is why I’m trying to keep both sides wet but minimize the volume on 1 side.
I’ve tried to grow cells at an air-liquid interface but they tend not to proliferate (a lung epithelial cell would grow in that set-up).