NP + IgG cleanup in 0.1% Tween 20
Our previous work with reverse centrifugation NPN membranes has shown that they may work for cleaning up excess protein from conjugated NPs. This initial success used ~20 nm red fluospheres with membranes from wafer 1070 (10-15%, ~45nm) and unlabeled IgG we were given by a collaborator. In this post we are testing “newer” NPN wafers that have larger porosity and pore size. Specifically, 1081(27%,75nm) and 1085 (26%,70nm).
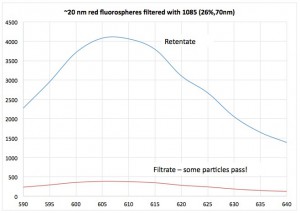
Mary Lee and I started out by testing a filter from 1085 with 20 nm red NPs in PBS/pixy that we had used in previous experiments. In the following fluorescence plot we show that some of the reds seem to be making it past our membrane.
Jumping ahead a little, we also tested a solution of 20nm reds in Tween 20 with a commercial Nanosep 300k filter. The NPs clearly went through by just a visual test:

We also own some blue NPs that measure slightly larger than the reds according to the Malvern. So we wanted to try them out to see if their slightly larger size would be enough make a difference with wafers 1081 and 1085.

In the rest of this post we continue exploring reverse centrifugation using the latest NPN wafers and Tween 20 as a surfactant. (We have also recently shown in preliminary tests that Tween 20 may be used to keep a combination of polystyrene fluospheres and IgG in solution.) All solutions are made using 0.1% Tween 20 in 1XPBS. Unless otherwise indicated, this post focussed on ~20 nm “blue” fluospheres and fluorescently tagged yellow IgG. Specifically, we are comparing our membranes to commercial filters. Our NPN membrane pore sizes fall roughly in between standard commerical filters with molecular weight cut-offs of 100kD and 300 kD. Here’s a nice table I found to help put the MWCOs in perspective:
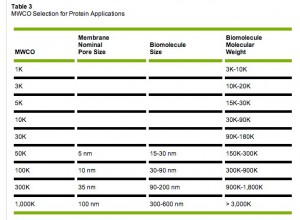
For this post we only had available Pall Nanosep 300kD filters and Millipore Microcon 100kD filters. (We also have an abundance of Microcons at 50kD, 10kD etc…)
You can find some details on the Nanosep filters here or here: Pall(Nanosep)
Nanosep Devices, Filter Media: Omega (modified polyethersulfone) ultrafiltration membrane, Sample Reservoir, Membrane Support Base, and Filtrate Receiver: Polypropylene
And Microcon info is here or here: Millipore(Microcon)
Interestingly, they no longer sell a “100k” MWCO filter. They are simply called “for Protein and DNA concentration”. The filter material is regenerated cellulose.
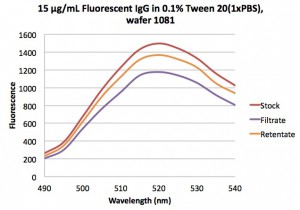
We started by using the 20 nm blues and IgG separately. This data looks great! The IgG passes freely and the 20 nm blues are completely blocked!
Next we put 20 nm blues in the same filter:
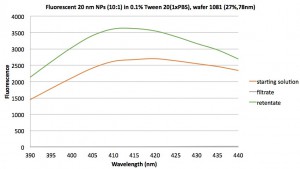
Because we are more interested in clean-up, we need to look at the NPs and IgG combined. So we made a solution of 10:1 diluted NPs and ~25 µg/mL IgG in 0.1%Tween 20. We put this solution in the same filter used above, as well as Nanosep and Microcon filters. That data along with some serial dilutions is presented in the next plot:

Long story short, our membranes are blocking 20 nm blues better than Nanosep 300 k filters and passing IgG better than Microcon 100k filters. Unfortunately, we hoped to look much better. Or at least, we thought they would look worse. As Jim immediately pointed out, the Microcon literature claims a 100k filter should stop 95% of the IgG. So we have some more thinking/work to do.