BSA concentration has extreme and significant effect on rate of diffusion through membrane
For a while now I’ve been running diffusion experiments using bacterial alkaline phosphatase (bAP) as my solute of interest in a solution of 50mM tris buffered saline (TBS) and 20 ug/mL BSA. Initially, the BSA was included as a way of reducing the adsorption of the low concentrations of bAP to the device surfaces — this specific concentration was chosen as roughly the lowest value that would still roughly maximize the protein’s effectiveness in reducing adsorption, and no change has been made to this concentration over the course of the vast majority of my experiments over the past year.
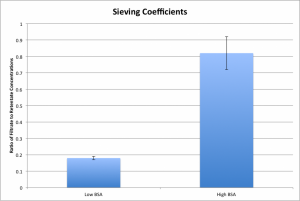
Recently, I made a mistake during my procedure and forgot a dilution step when preparing the BSA, instead using a concentration of 93 ug/mL in an otherwise normal 1hr 100um gasket, bottom-to-top covered diffusion test. The results were astonishing.
The high BSA concentration drove the system nearly to equilibrium over the same amount of time, whereas the low concentration system is still below a sieving coefficient value of 0.2. This is repeatable: n = 3 for both of these values. In order to ensure that everything checked out, I went through and calculated the mass of bAP that was observed in each of the wells.
This looks precisely as expected if the high BSA concentration is assumed to accelerate diffusion — no significant difference exists between the total mass, but the high BSA case has a significantly higher amount of bAP in the top well (the filtrate) and a significantly lower amount in the bottom well (the retentate.)
There’s no (obvious) justification for this behavior presented in the literature. BSA is known to bind to and carry lipids in water; to coat hydrophobic surfaces and present hydrophilic regions; and to attract and bind water. A quick calculation of the theoretical time-to-equilibirum for this system without accounting for BSA according to the equation t = (L^2)/2D where L is the largest diffusion distance and D is the diffusion coefficient suggests that it should take roughly 2.7 hours, indicating that the contribution of the increased BSA concentration is indeed to speed up diffusion, rather than simply not slowing it down. More investigation is required to understand and characterize this effect.

There is an equal concentration of BSA on both sides of the membrane, right?
That’s right; the only difference between the solutions of the two wells is the presence of 250 ng/mL initial concentration of alkaline phosphatase on the retentate side (the bottom well.)