Preliminary work on a membrane-bound biosensor
You may recall that last year I spent some time working on a proof-of-concept experiment for a high-throughput, non-specific drug discovery assay. The experiment involved binding up a FLAG-peptide sequence-tagged alkaline phosphatase (AP) enzyme with a biotinylated anti-FLAG antibody, in turn bound to a large avidin bead. This very large complex was unable to move across the nanoporous membrane unless it was disrupted by free FLAG peptide added to the solution, which eluted the enzyme off of the antibody, allowing it to diffuse into the filtrate for detection via a fluorescein diphosphate (FDP) substrate.
That work was a success, but now we’ve decided to repurpose the same biochemistry for a different application. Recently, we have developed a nanoporous “tent” which can be overlaid over a chip surface for two membranes in close series (see Greg’s post, here.) One potential application for this type of device is similar in theory to the drug discovery assay concept we had pursued previously: a biosensor bound to the chip surface could be buffered from the bulk fluid by the tent membrane, and its substrate bound up in a complex too large to diffuse through the tent to see the sensor. Disruption of the complex would then free up the substrate to enter the space between the tent and the chip and interact with the sensor.
To test this concept, a simple assay must be developed. As our model biosensor, we will use the fluorescent substrate to our enzyme, FDP, bound to the chip surface; as our analyte, the enzyme itself, AP. This will allow binding of the enzyme to a larger AP-antibody-avidin complex later on; however, for now, we simply want to confirm that the chip surface can be bound to FDP and that addition of AP to these chips will result in a visible fluorescent signal.
To bind FDP to the surface of the chip, it was hypothesized that incubation with Poly-L-Lysine (PLL) may help hold FDP to the surface via ionic interaction between the highly positive PLL and the highly negative FDP. Four chips were prepared:
| Treated with AP | No AP | |
| Treated with PLL | PLL-AP | PLL-Sham |
| No PLL | Sham-AP | Sham-Sham |
Incubation with PLL occurred in DI water over roughly two hours at room temperature, before rinsing three times with DI. Chips not treated with PLL were incubated only in DI. All four chips were then incubated with FDP in 1X PBS for an hour before rinsing with PBS three times. Alkaline phosphatase added to the appropriate chips was allowed to sit for five minutes to allow the signal to develop before measurement, which was performed by fluorescence microscopy using a FITC excitation laser. Sample images recovered for each chip are shown:
| Treated with AP | No AP | |
| Treated with PLL |  |
 |
| No PLL |  |
 |
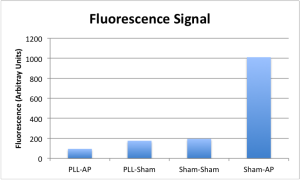
The results are visually quite clear, but quantification is offered by way of imageJ:
Quite the opposite of what we initially anticipated, the PLL treatment has an obvious negative effect on the availability of FDP to AP — either because it doesn’t bind the FDP as well as bare membrane, or more likely, because it binds it so well that the AP is unable to access its substrate well enough for dephosphorylation to occur. Either way, this gets us where we wanted to go in a more direct manner: FDP incubated on the surface on its own sticks well enough to create a clear boost in signal when the analyte is made available to it.
The next direction to take this involves binding up the AP with a large complex by interaction with the antibody-avidin pair, and then demonstrating that fluorescence is still significantly increased when the large complex is available. Once the complex is shown to be active, the final step is to show that addition of the tent membrane turns off this activity, and that disruption of the complex restores it.