Urea Clearance: First Animal Study Results
BOTTOM LINE: We had great results from the small number of nanoporous membranes used to clear urea from Sprague-Dawley rats. We are clearing Urea better than commercial materials. (see fig)
Recap of previous work:
First we leaned how to set up the dialysis experiments using Sprague-Dawley rats.
We made some rats uremic to elevate the uremia levels.
Then we did sham dialysis with non-porous nitride membranes.
The Studies with non-porous membranes showed that we could raise the urea level of the animals and perform four hours of dialysis without distressing the animals. (beyond making them uremic, which brings on weight loss and causes the animals to look ‘ruffled’)
We then dialyzed rats using commercial membranes of both Polyethersulfone (PES)and Cellulose triacetate (CTA) and nanoporous nitride membranes from SiMPore. These chips had the narrow slots and no vias. As a reminder, blood is pumped from the femoral artery, via a peristaltic pump, through our membrane device, and back to the animal by way of the femoral vein. Blood samples are taken from the femoral artery at the start then every hour for the four hour treatment/experiment. The urea levels measured by the colormetric assay is the blood urea level of the animal, not the urea in the blood exiting the dialysis device. This is how we determine KT/V which equals ln(C0/C), where C0 is the initial urea concentration and C is the current urea concentration.
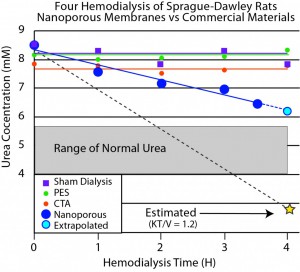
The graph may be clearer if I normalized each membrane type by the starting urea concentration, but it is evident that the sham dialysis, purple, and both commercial membranes, green (PES) and orange (CTA) had no affect on the urea concentration in the animal. The nanoporous membranes, blue, on the other hand reduced the urea substantially. There was a problem with the mambrane at 3.5 hours and the hour four data is extrapolated. The dotted line to the star shows the expected clearance if the area used for the commercial membranes had been used for the nanoporous membranes. This would give us the clearance of a typical in-center dialysis treatment, KT/V = 1.2.
This is an excellent summary and a historic moment for us.
After years of thinking about this result theoretically, we have real evidence of the benefits our our membranes over commercial dialysis membranes for clearing toxins from blood.
I may be benefiting us with some rounding errors, but my take home message is that we need 100x less membrane to clear blood than conventional dialysis membranes. This is like going from the area of a twin mattress to a couple of index cards. Lift-off is indeed the key to making such miniaturization truly useful.
Congratulations on obtaining the first true hemodialysis data on our nanoporous material!!
I assume this is Milestone #1, so I’m curious what Milestone #2 will be? How far do you need to get to justify moving forward to larger animal studies? What are the limitations that need to be overcome?
Regarding the comparative commercial membranes, are these the exact same membranes that are found inside dialyzers used for human Hemodialysis or are they lab dialysis membranes? I assume that the hollow fiber membranes inside of dialyzers are much different than the membranes used in lab dialysis, so understanding any differences will be important.
Regarding liftoff, if we had a large sheet of perfect lifted off material today, do we have a vision of what we would do with it? Have any prototypes been made with the microporous material that has already been lifted off? Hopefully the current on-wafer designs can generate the type of data that is needed for the next several milestones.
Thanks for sharing the good news!
Thanks for the summary! And congrats on the positive results!
Were we able to get data showing that we didn’t lose too much of the good parts of blood along with the Urea? Can you measure the dialysate for this? I ask because I assume we could see the same results if we used one of our 350 nm porous membranes or any pore size that is smaller than red blood cells but larger than Urea. I know these pore sizes were not optimal, but even showing that there was a slight differentiation in diffusion of Urea vs Albumin would be interesting. (I think Tucker has already shown that we would expect there to be a difference in his diffusion tests?)
Thanks! – Josh
The commercial membranes used are the same material, but not the same form factor used in HD. The objective was to compare materials with the same form factor, flat sheets not bundles of hollow tubes.
We have started thinking about what do to with lift-off material. That is one of the things we will focus on before we begin our next rat study.
We did not check Albumin loss in this study. We’ve done that in bench top experiments with serum. This is certainly something we will keep in mind when designing our next animal study, including removal of larger molecules like ß2-µglobulin.