ADSC Endothelial Differentiation Manuscript
Membranes Promote Endothelial Differentiation of Adipose-Derived Stem Cells and Perivascular Interactions
Submitted to Journal of Cellular and Molecular Bioengineering (a BMES journal)
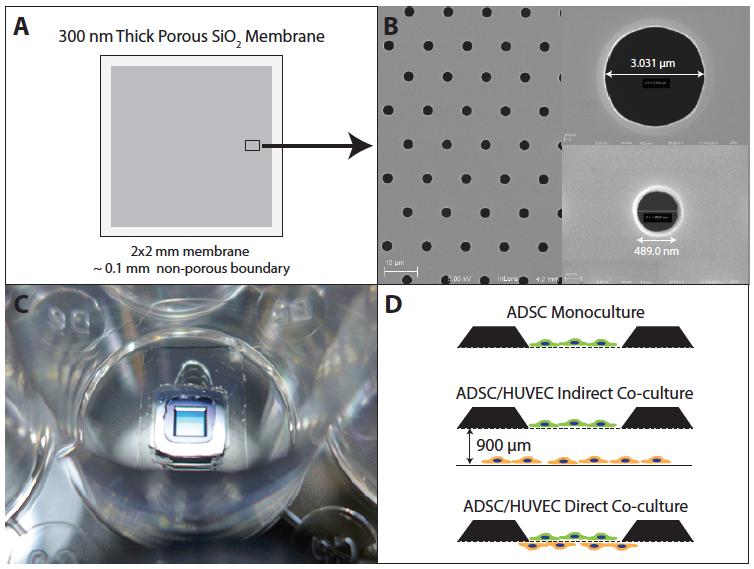
FIGURE 1. Ultrathin microporous silicon dioxide (SiO2) membranes. (A) 300 nm thick SiO2 membranes were fabricated on silicon wafers and etched into 5.4 x 5.4 mm dies with 2 x 2 mm membrane windows. Within the suspended membrane, pores were patterned inside of an approximately 100-micron exclusion zone. (B) Pores were etched in a hexagonal tiling pattern with center-center pore spacing of 4 diameters. Membranes were patterned with either 3.0 or 0.5 micron diameter pores. (C) Membrane chips were bonded to silicone gaskets, which were then bonded to the bottom of 24-well plates. (D) Membranes were used for either ADSC monocultures, indirect co-culture with HUVEC or direct co-cultures.
FIGURE 2. ADSCs migrate to and proliferate over porous regions of membranes. 3.0 and 0.5 micron pore SiO2 membranes were pre-coated with a 1% Geltrex™ solution. ADSC were seeded at an approximate density of 1000 cells per membrane at day 0. ADSC seeded on 3.0 micron (A) or 0.5 micron (B) pore diameter SiO2 membranes and grown in complete endothelial media supplemented with 50 ng/mL VEGF for 6 days. ADSCs appeared to migrate and proliferate primarily over the porous region of the suspended membrane.
FIGURE 3. ADSC expression of PECAM-1 (CD31) after culture on membranes in different media. ADSC were cultured for 6 days in three different media conditions prior to fixation and staining. (A) ADSCs cultured in complete EC media with 50 ng/mL of VEGF expressed a relatively uniform distribution of CD31. (B) ADSCs cultured in basal EC media without growth factors expressed a lower uniform distribution of CD31. (C) CD31 expression was negligible when ADSCs were cultured in proliferation media.
FIGURE 4. Indirect co-culture of ADSCs with HUVECs results in endothelial-like expression of CD31. ADSCs were cultured for 6 days on 3.0 micron pore SiO2 membranes while HUVECs were indirectly co-cultured on the bottom of the 24-well plate. (A) In complete EC media with 50 ng/mL VEGF, a subpopulation of ADSC displayed an endothelial-like surface expression of CD31 (see Figure S2), while remaining ADSC expressed a low-level uniform distribution of CD31 (similar to Figure 3a). (B) Indirect co-culture of ADSC and HUVEC in basal EC media did not result in significant expression of CD31.
FIGURE 5. ADSCs differentiated on porous membranes produce more branch points and greater overall tube lengths in the standard angiogenesis assay. ADSCs were cultured in either complete EC media with 50 ng/mL VEGF or basal EC media. Within each media group, cells were either cultured on 3.0 micron high-porosity track-etched membrane inserts or tissue culture treated 24-well plates. All surfaces were pre-treated with a 1% Geltrex™ solution. After 6 days, cells were harvested and counted. 2500 cells were seeded on top of a Geltrex™ gel in a 96-well plate. After 18 hours, multiple images of each well were taken at 4x. The images were reconstructed to produce a composite image of the entire well (Figure S3). Branch points (A) and total tube length (B) were measured. In all cases, ADSCs differentiated on membranes generated significantly more branch points and greater total tube length. Representative images of the wells are shown: (C) complete EC media with 50 ng/mL VEGF on membranes, (D) basal media on membranes, (E) complete EC media with 50 ng/mL VEGF on TCP, and (F) basal media on TCP. Sample size n=6 for each condition. Error bars are SD. Statistical comparisons are unpaired student t-test assuming equal variance.
FIGURE 6. ADSCs orient perpendicularly to HUVECs across a 0.5 micron pore size SiO2 membrane. HUVECs and ADSCs were directly co-cultured across a SiO2 membrane in complete EC media with 50 ng/mL VEGF. (A) HUVECs were seeded at high density and cultured to confluence on the underside of the membrane prior to seeding ADSCs on the top surface. (B) 1 day after seeding ADSCs, some of the ADSCs became elongated and began to orient perpendicularly to the HUVECs (arrowheads). (C) After 3 days of co-culture, the HUVECs formed semi-organized patterns, similar to monocultures of HUVECs in the presence of VEGF (Figure S2). (D) At 6 days, the majority of the ADSCs oriented perpendicularly to the HUVECs. Box outlines indicate the location of enlarged images (E-H).
FIGURE 7. Undifferentiated ADSC orient perpendicularly to differentiated ADSC. ADSCs were indirectly co-cultured with HUVECs on 3.0 micron pore SiO2 membranes in complete EC media with 50 ng/mL VEGF for 6 days. A subpopulation of the ADSC began to display endothelial-like CD31 expression at the cell boundaries, while the remainder of the cells expressed a low-level uniform distribution of CD31. (A) The population of ADSC displaying high levels of CD31 had predominately migrated through the 3.0 micron pores to the backside of the membrane. (B) Imaging of the ADSCs above the membrane indicated that the majority of these cells expressed low levels of CD31 in a uniform distribution. These undifferentiated cells oriented perpendicularly to the differentiated ADSCs. Dotted lines show the orientation of the cells based on the polarization of the nucleus (DAPI) and CD31.
FIGURE S1. ADSC expression of CD31 after culture on tissue culture treated plastic in different media. Wells of a 24-well plate were pre-coated with a 1% Geltrex™ solution. ADSC were cultured for 6 days in three different media conditions prior to fixation and staining. (A) ADSCs cultured in complete EC media with 50 ng/mL of VEGF expressed a relatively uniform distribution of CD31. (B) ADSCs cultured in basal EC media without growth factors expressed a lower uniform distribution of CD31. (C) CD31 expression was negligible when ADSCs were cultured in proliferation media.
FIGURE S2. Monocultures of HUVEC develop random orientations when cultured in complete EC media with VEGF. HUVEC were cultured on 1% Geltrex™ coated tissue culture treated plastic until reaching confluence. (A,B) HUVEC were fixed and stained for nuclei (DAPI) and actin (phalloidin-AlexaFluor 594). (C,D) CD31 expression (green) was predominantly localized to the cell boundaries.
FIGURE S3. Image reconstructions of the 96-well plate angiogenesis assay. In order to count all branch points and total tube length in each well of the angiogenesis assays, multiple 4x bright field images were stitched together using Adobe Photoshop. Representative images are shown of ADSC differentiated in the following conditions prior to the angiogenesis assay: complete EC media with 50 ng/mL VEGF on membranes (A), basal EC media on membranes (B), proliferation media on membranes (C), complete EC media with 50 ng/mL VEGF on TCP (D), basal EC media on TCP (E) and proliferation media on TCP (F).